Supercritical Fluid Co2
Supercritical Fluid Examples Properties And Phase Diagrams Supercritical carbon dioxide (sco 2[disputed – discuss]) is a fluid state of carbon dioxide where it is held at or above its critical temperature and critical pressure. Supercritical co2 (sc co 2) with its reduced viscosity, increased diffusivity, and liquid like density, is attractive for enhanced hydrocarbon recovery, shale gas fracturing and drilling (especially in underbalanced condition).
Supercritical Fluid Examples Properties And Phase Diagrams The effect of long range order parameter fluctuations on the thermodynamic and transport properties of supercritical fluids (sc co2) will be discussed. When carbon dioxide is heated and compressed past this threshold, it becomes a single, homogeneous phase known as a supercritical fluid. the conditions required to transform co2 into this state are relatively mild compared to other substances, which is a major factor in its industrial practicality. Supercritical carbon dioxide (sco2) emerges as an effective working fluid in closed loop energy conversion cycles, offering significant advantages over traditional steam based rankine cycles. In this perspective, we review microfluidic investigations involving supercritical co 2 , with a particular focus on three primary applications, namely, solvent extraction, nanoparticle synthesis, and chemical reactions.

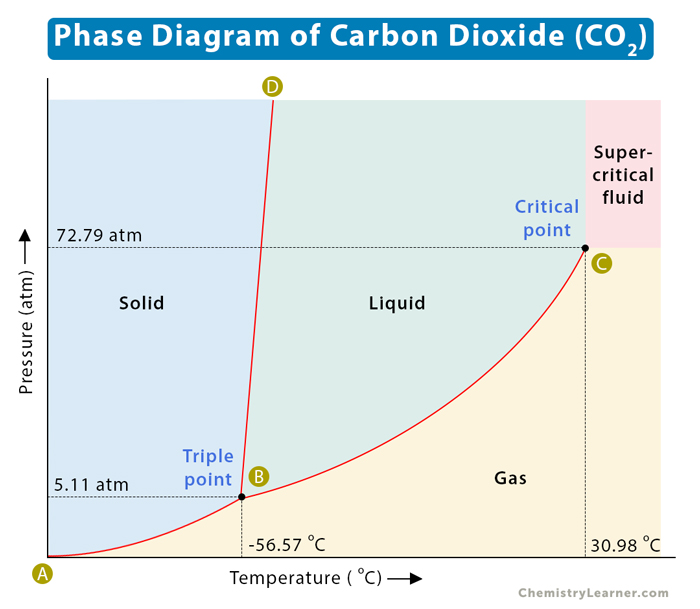
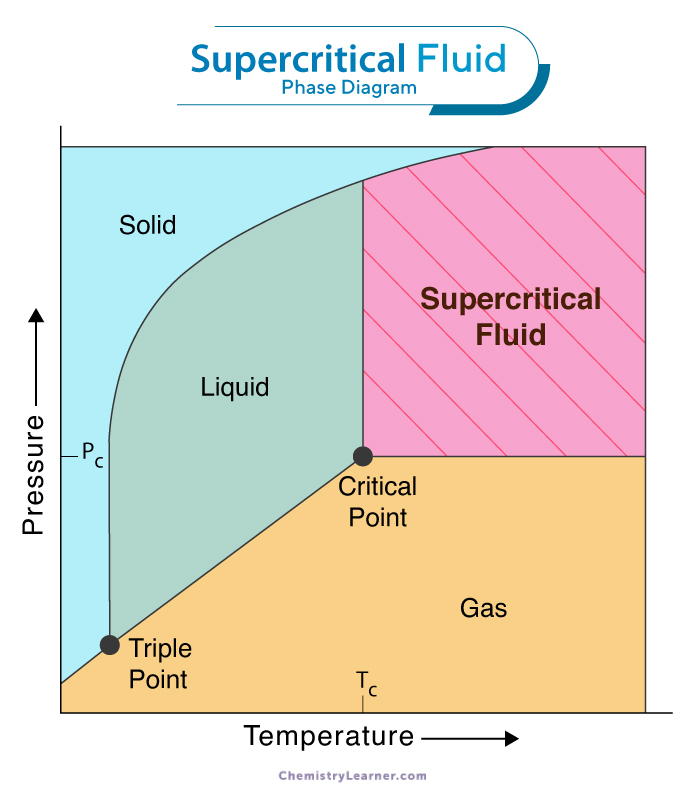
Supercritical Fluid Extraction Supercritical Co2 Extraction Supercritical carbon dioxide (sco2) emerges as an effective working fluid in closed loop energy conversion cycles, offering significant advantages over traditional steam based rankine cycles. In this perspective, we review microfluidic investigations involving supercritical co 2 , with a particular focus on three primary applications, namely, solvent extraction, nanoparticle synthesis, and chemical reactions. This paper presents a comprehensive review of all correlations and experimental studies available in the literature to determine the heat transfer coefficient of supercritical co2 flowing in heat exchangers. the different applications in which it is used are also reviewed and discussed. Supercritical carbon dioxide (co 2) is a state of carbon dioxide where it is held at or above its critical temperature and critical pressure. carbon dioxide transitions into a supercritical fluid at temperatures above 31.1°c (88°f) and pressures above 73 atmospheres (1071 psi). The present review provides comprehensive analysis of the thermodynamic and transport properties of supercritical carbon dioxide and co 2 containing binary mixtures (experiment and theory) and their various technological and scientific applications in different natural and industrial processes. Due to its high availability and low toxicity, as has been already mentioned, the most widely used supercritical fluid is carbon dioxide, although its use in many processes is limited by the low solubility of many reactants and products, even in supercritical conditions.

Supercritical Fluid Diagram This paper presents a comprehensive review of all correlations and experimental studies available in the literature to determine the heat transfer coefficient of supercritical co2 flowing in heat exchangers. the different applications in which it is used are also reviewed and discussed. Supercritical carbon dioxide (co 2) is a state of carbon dioxide where it is held at or above its critical temperature and critical pressure. carbon dioxide transitions into a supercritical fluid at temperatures above 31.1°c (88°f) and pressures above 73 atmospheres (1071 psi). The present review provides comprehensive analysis of the thermodynamic and transport properties of supercritical carbon dioxide and co 2 containing binary mixtures (experiment and theory) and their various technological and scientific applications in different natural and industrial processes. Due to its high availability and low toxicity, as has been already mentioned, the most widely used supercritical fluid is carbon dioxide, although its use in many processes is limited by the low solubility of many reactants and products, even in supercritical conditions.

Supercritical Fluid Diagram The present review provides comprehensive analysis of the thermodynamic and transport properties of supercritical carbon dioxide and co 2 containing binary mixtures (experiment and theory) and their various technological and scientific applications in different natural and industrial processes. Due to its high availability and low toxicity, as has been already mentioned, the most widely used supercritical fluid is carbon dioxide, although its use in many processes is limited by the low solubility of many reactants and products, even in supercritical conditions.

Supercritical Fluid Diagram
Comments are closed.