Solved Question 2 1 Pts Which Reason Explains Why Gas Bubbles Are
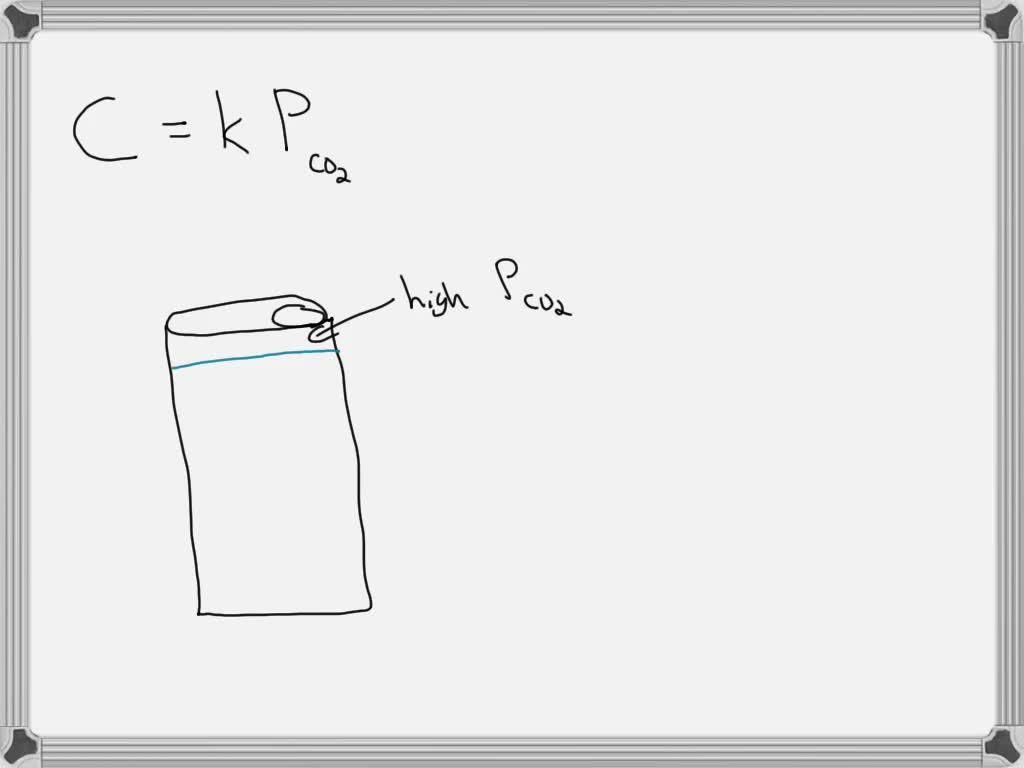
Solved Question 2 1 Pts Which Reason Explains Why Gas Bubbles Are Question: question 2 1 pts which reason explains why gas bubbles are released when a carbonated beverage is opened? the solvent changes from a liquid to a gas when the beverage is opened. The gas bubbles in a carbonated beverage are released when opened because the solubility of the gas decreases with decreased pressure. when the pressure drops, the liquid can no longer hold as much carbon dioxide, causing it to escape as bubbles.

Question 2 1 Pts Which Reason Explains Why Gas Bubbles Are Released When the pressure is reduced, the carbon dioxide is released from the solution as small bubbles, which causes the solution to become effervescent, or fizzy. a common example is the dissolving of carbon dioxide in water, resulting in carbonated water. Video answer: the solubility of a gas in a solvent can be described by henry's law, where the concentration or solubility is equal to a constant multiplied by the vapor pressure of the gas above the solution, in this case, carbon dioxide. Which reason explains why gas bubbles are released when a carbonated beverage is opened. A cloud is like the bubbles in a soft drink, but in reverse. a cloud is made up of small water droplets in air, whereas the bubbles are just gas droplets in a liquid. both water droplets and bubbles require nucleation sites to form. you can also observe it in a cloud chamber in the laboratory.

Explain Why Gas Bubbles Rise To The Surface Of A Glass Of Soda Which reason explains why gas bubbles are released when a carbonated beverage is opened. A cloud is like the bubbles in a soft drink, but in reverse. a cloud is made up of small water droplets in air, whereas the bubbles are just gas droplets in a liquid. both water droplets and bubbles require nucleation sites to form. you can also observe it in a cloud chamber in the laboratory. When you open a bottle or can, the pressure above the carbonated liquid drops to match the pressure outside of the bottle or can. the pressure release results in a hissing sound, and you see. Gas bubbles form because the solubility of carbon dioxide decreases as the soda warms up, causing the gas to escape and form bubbles. The bubbles of gas form in a glass of soda water when it warms up because gases are less soluble in warm liquids than in cold liquids. when the soda water warms up, the solubility of the gas (usually carbon dioxide) decreases, causing the gas to escape from the solution in the form of bubbles. Bubbles are more than just simple pockets of air in water. their formation is governed by several scientific principles, explaining how and why these gaseous inclusions emerge within a liquid. understanding these mechanisms reveals the dynamic interactions between water, dissolved gases, and energy.
Comments are closed.