Gas Laws Question 2

Gas Laws Diagram Quizlet A container containing 5.00 l of a gas is collected at 100 k and then allowed to expand to 20.0 l. what must the new temperature be in order to maintain the same pressure (as required by charles' law)?. According to boyle's law, the volume of a gas is inversely proportional to its pressure, when the temperature and amount of gas are constant. therefore, if the pressure decreases by a factor of 5 (from 5.0 atm to 1.0 atm), the volume will increase by the same factor.

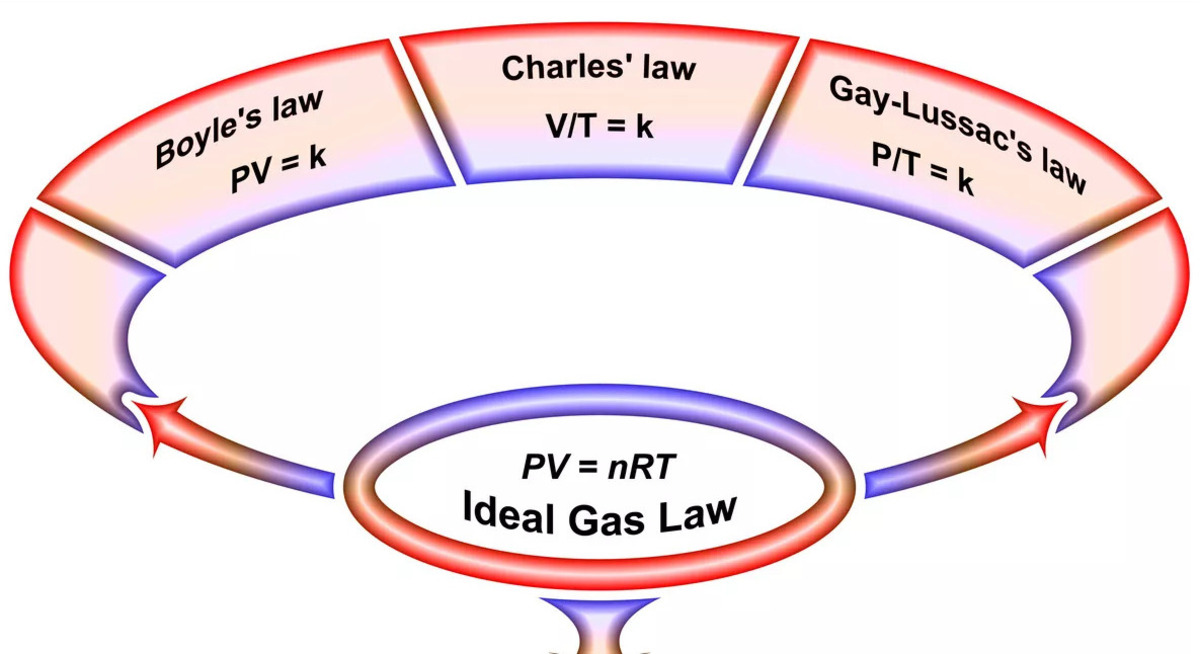
Gas Laws Worksheet The document is a practice test containing 25 multiple choice questions about gas laws and the behavior of gases. the questions cover topics like how changing temperature, pressure, and volume affect gases based on the gas laws (e.g. boyle's law, charles's law, ideal gas law). The following practice problems are to master to topics on the ideal gas laws: boyle’s law, charles’s law, and avogadro’s law, as well as the combined gas law equation. there are examples to work on the dalton law of partial pressures, the graham’s law of effusion, and gas stoichiometry. In 1809, joseph louis gay lussac gave the gay lussac's law or the pressure law also known as amontons' law. according to this law, at constant volume, the pressure of a fixed amount of gas varies directly with temperature. We have 17 ready to use problem sets on the topic of gas laws and gas stoichiometry. these problem sets will focus on the mathematical relationship and inter dependency between the pressure, volume, temperature, and the number of moles of a sample of gas.

Gas Laws Worksheet 1 Key Pdf Worksheets Library In 1809, joseph louis gay lussac gave the gay lussac's law or the pressure law also known as amontons' law. according to this law, at constant volume, the pressure of a fixed amount of gas varies directly with temperature. We have 17 ready to use problem sets on the topic of gas laws and gas stoichiometry. these problem sets will focus on the mathematical relationship and inter dependency between the pressure, volume, temperature, and the number of moles of a sample of gas. Solve these gas laws questions and sharpen your practice problem solving skills. we have quizzes covering each and every topic of physical chemistry and other concepts of chemistry. What are the gas laws. how many are there. learn their types, statements, and formula. also, learn how to do and solve gas law problems. As temperature of a gas increases, pressure will also increase based on the ideal gas law. the volume of the tire can only expand so much before the rubber gives and releases the build up of pressure. A sample of unknown compound gas x is shown by analysis to contain sulphur and oxygen. the gas requires 28.3 seconds to diffuse through a small aperture into a vacuum.

Combined Gas Laws Worksheet Exercises Sections 10 3 10 4 The Gas Solve these gas laws questions and sharpen your practice problem solving skills. we have quizzes covering each and every topic of physical chemistry and other concepts of chemistry. What are the gas laws. how many are there. learn their types, statements, and formula. also, learn how to do and solve gas law problems. As temperature of a gas increases, pressure will also increase based on the ideal gas law. the volume of the tire can only expand so much before the rubber gives and releases the build up of pressure. A sample of unknown compound gas x is shown by analysis to contain sulphur and oxygen. the gas requires 28.3 seconds to diffuse through a small aperture into a vacuum.
8 Extraordinary Facts About Gas Laws Facts Net As temperature of a gas increases, pressure will also increase based on the ideal gas law. the volume of the tire can only expand so much before the rubber gives and releases the build up of pressure. A sample of unknown compound gas x is shown by analysis to contain sulphur and oxygen. the gas requires 28.3 seconds to diffuse through a small aperture into a vacuum.
Comments are closed.