Solution Auger Effect Studypool

Auger Effect Pdf Two views of the auger process. (a) illustrates sequentially the steps involved in auger deexcitation. an incident electron (or photon) creates. Auger effect, in atomic physics, a spontaneous process in which an atom with an electron vacancy in the innermost (k) shell readjusts itself to a more stable state by ejecting one or more electrons instead of radiating a single x ray photon. this internal photoelectric process is named for the french physicist pierre victor auger, who discovered it in 1925. (however, the effect had been.

Solution Auger Effect Studypool Abstract this work presents an essay to the auger effect, by the inelastic collision theory. calcula tions of the energies of the electrons ejected for bands of the auger spectrum of some mo lecules were made, to test of the model. The auger effect is named after its discoverer, pierre auger, who observed a tertiary effect while studying photoemission processes in the 1920s. auger electrons are emitted at discrete energies that allow the atom of origin to be identified. the idea of using electron stimulated auger signals for surface analysis was first suggested in 1953 by j. j. lander. the technique became practical for. The auger effect is defined as the emission of secondary electrons from an atom or molecule due to the electron electron interaction in the presence of an inner shell vacancy, where an outer shell electron recombines into the vacant orbital, providing enough energy to ionize another outer shell electron. this phenomenon serves as a basic manifestation of electronic correlation and can be. Today’s lecture: auger effect (radiation less transitions) and moseley’s law you must be able to understand auger effect and moseley’s plot. 1 f history pierre auger, in 1925 observed (at first in the cloud chamber, then in photographic plates) the occurrence of electrons with precisely determined energies. these electrons have been later named auger electrons) may serve to identify.

Auger Process Auger Effect Wikipedia Ionization Energy Quantum The auger effect is defined as the emission of secondary electrons from an atom or molecule due to the electron electron interaction in the presence of an inner shell vacancy, where an outer shell electron recombines into the vacant orbital, providing enough energy to ionize another outer shell electron. this phenomenon serves as a basic manifestation of electronic correlation and can be. Today’s lecture: auger effect (radiation less transitions) and moseley’s law you must be able to understand auger effect and moseley’s plot. 1 f history pierre auger, in 1925 observed (at first in the cloud chamber, then in photographic plates) the occurrence of electrons with precisely determined energies. these electrons have been later named auger electrons) may serve to identify. Radiative auger satellites (ras) •the radiative auger effect gives rise to satellites in characteristic x ray spectra •satellites are features surrounding the main transition profile. Auger effect the auger effect is a process by which electrons with characteristic energies are ejected from atoms in response to a downward transition by another electron in the atom. in auger spectroscopy, the vacancy is produced by bombardment with high energy electrons, but the auger effect can occur if the vacancy is produced by other interactions. it is observed as one of the methods of. The fluorescence and auger effect through these two phenomena are not directly related with the nuclear phenomena, they appear as a consequence of some nuclear event, i.e. orbital capture and internal conversation, leading to a vacancy in the electron orbitals, usually in the k or l shell. to fill in such energetically unstable vacancies, the electrons jump from the outer orbitals in cascade. Auger effect the auger effect is a physical phenomenon in which the filling of an inner shell vacancy of an atom is accompanied by the emission of an electron from the same atom. [1] when a core electron is removed, leaving a vacancy, an electron from a higher energy level may fall into the vacancy, resulting in a release of energy.
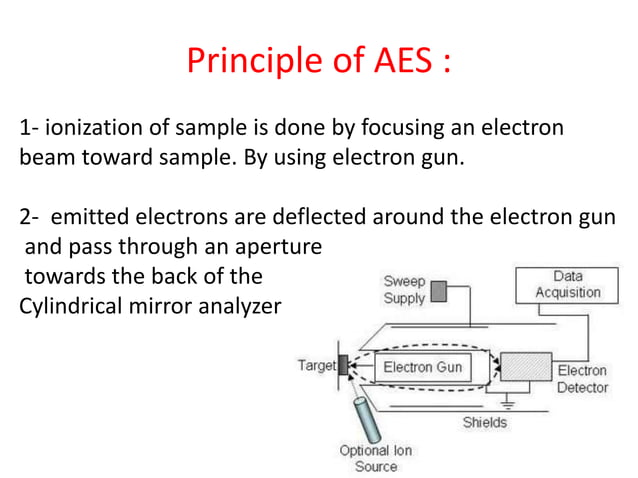
Auger Effect Puplic Pptx Radiative auger satellites (ras) •the radiative auger effect gives rise to satellites in characteristic x ray spectra •satellites are features surrounding the main transition profile. Auger effect the auger effect is a process by which electrons with characteristic energies are ejected from atoms in response to a downward transition by another electron in the atom. in auger spectroscopy, the vacancy is produced by bombardment with high energy electrons, but the auger effect can occur if the vacancy is produced by other interactions. it is observed as one of the methods of. The fluorescence and auger effect through these two phenomena are not directly related with the nuclear phenomena, they appear as a consequence of some nuclear event, i.e. orbital capture and internal conversation, leading to a vacancy in the electron orbitals, usually in the k or l shell. to fill in such energetically unstable vacancies, the electrons jump from the outer orbitals in cascade. Auger effect the auger effect is a physical phenomenon in which the filling of an inner shell vacancy of an atom is accompanied by the emission of an electron from the same atom. [1] when a core electron is removed, leaving a vacancy, an electron from a higher energy level may fall into the vacancy, resulting in a release of energy.

Auger Effect The fluorescence and auger effect through these two phenomena are not directly related with the nuclear phenomena, they appear as a consequence of some nuclear event, i.e. orbital capture and internal conversation, leading to a vacancy in the electron orbitals, usually in the k or l shell. to fill in such energetically unstable vacancies, the electrons jump from the outer orbitals in cascade. Auger effect the auger effect is a physical phenomenon in which the filling of an inner shell vacancy of an atom is accompanied by the emission of an electron from the same atom. [1] when a core electron is removed, leaving a vacancy, an electron from a higher energy level may fall into the vacancy, resulting in a release of energy.

Auger Effect Puplic Pptx
Comments are closed.