Auger Effect

Auger Effect Pdf The auger effect can impact biological molecules such as dna. following the k shell ionization of the component atoms of dna, auger electrons are ejected, leading to damage to its sugar phosphate backbone. Auger effect, in atomic physics, a spontaneous process in which an atom with an electron vacancy in the innermost (k) shell readjusts itself to a more stable state by ejecting one or more electrons instead of radiating a single x ray photon.
Auger Effect Alchetron The Free Social Encyclopedia Auger electron spectroscopy (auger spectroscopy or aes) was developed in the late 1960's, deriving its name from the effect first observed by pierre auger, a french physicist, in the mid 1920's. When an auger electron is emitted as a result of an electron transition from a higher subshell to a lower subshell of the same shell, it is referred to as a coster–kronig transition, which is a special type of auger effect named in honor of dirk coster and ralph de lear kronig from the netherlands. Learn about the auger effect, a phenomenon in which electrons are ejected from atoms instead of emitting x rays. find out how auger electron spectroscopy can be used to identify the emitting atoms and measure their composition. A review of auger radiotherapy, a targeted therapy based on the emission of low energy electrons from radioactive atoms. the article discusses the technological development, the potential carriers, and the clinical trials of auger emitters for cancer therapy.

26 Auger Effect Images Stock Photos Vectors Shutterstock Learn about the auger effect, a phenomenon in which electrons are ejected from atoms instead of emitting x rays. find out how auger electron spectroscopy can be used to identify the emitting atoms and measure their composition. A review of auger radiotherapy, a targeted therapy based on the emission of low energy electrons from radioactive atoms. the article discusses the technological development, the potential carriers, and the clinical trials of auger emitters for cancer therapy. These are the twenty auger peaks listed for these elemental solids in the national institute of standards and technology (nist) x ray photoelectron spectroscopy database. The auger effect occurs when an electron from an inner shell of an atom is removed, creating a vacancy. an electron from a higher energy level then fills this vacancy. Named after the french physicist pierre auger, who first discovered the phenomenon in the 1920s, the auger effect is a non radiative process in which an atom or molecule relaxes from an excited state by emitting an electron, known as an auger electron. A paper by e. h. s. burhop on the quantum mechanical calculation of the internal conversion coefficient for the auger effect, a radiationless transition of an electron in an atom. the author uses hydrogen like wave functions and compares the theoretical results with experimental data.
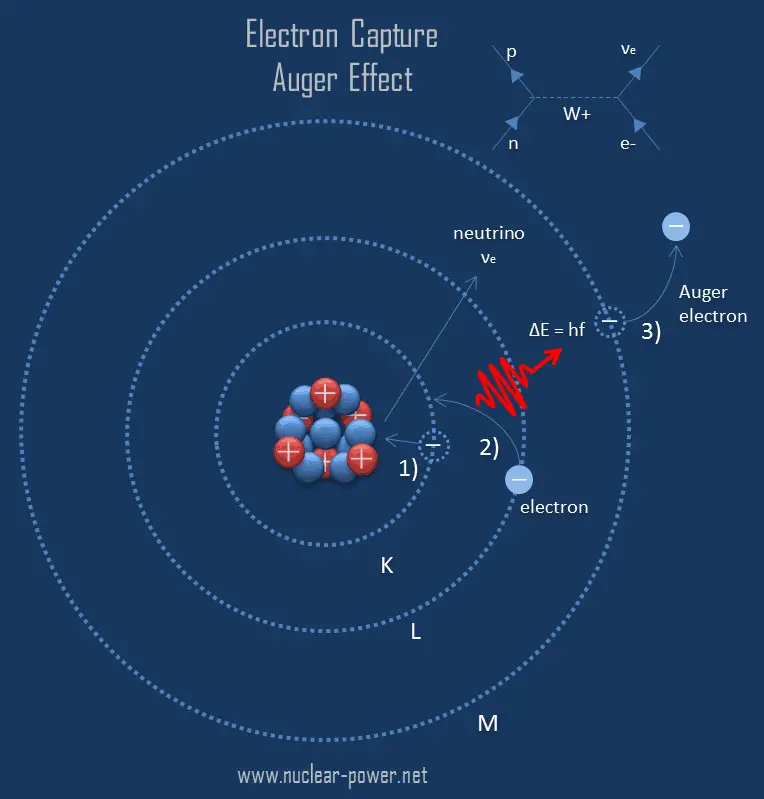
What Is Auger Effect Auger Electron Definition These are the twenty auger peaks listed for these elemental solids in the national institute of standards and technology (nist) x ray photoelectron spectroscopy database. The auger effect occurs when an electron from an inner shell of an atom is removed, creating a vacancy. an electron from a higher energy level then fills this vacancy. Named after the french physicist pierre auger, who first discovered the phenomenon in the 1920s, the auger effect is a non radiative process in which an atom or molecule relaxes from an excited state by emitting an electron, known as an auger electron. A paper by e. h. s. burhop on the quantum mechanical calculation of the internal conversion coefficient for the auger effect, a radiationless transition of an electron in an atom. the author uses hydrogen like wave functions and compares the theoretical results with experimental data.
Comments are closed.