Sn1 Sn2 E1 E2 Dont Guess Know Which Mechanism
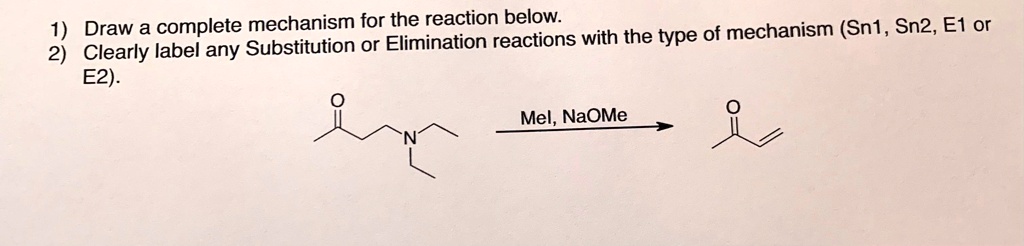
1 Draw A Complete Mechanism For The Reaction Below Of Mechanism Sn1 Sn2 Sn1 vs sn2 vs e1 vs e2: how to choose the right mechanism if you're taking organic chemistry, this is probably the topic keeping you up at night. an exam gives you a substrate, a nucleophile base, and a solvent, and you're supposed to predict whether the reaction runs sn1, sn2, e1, or e2. sometimes it's a mix. sometimes there's no reaction at all. there are four things to check, in order. If you haven’t seen my videos on the sn1, sn2, e1, and e2 mechanisms, i strongly encourage you to first watch those and then come back to this video. so, let’s start by a quick review of the important features of each mechanism.
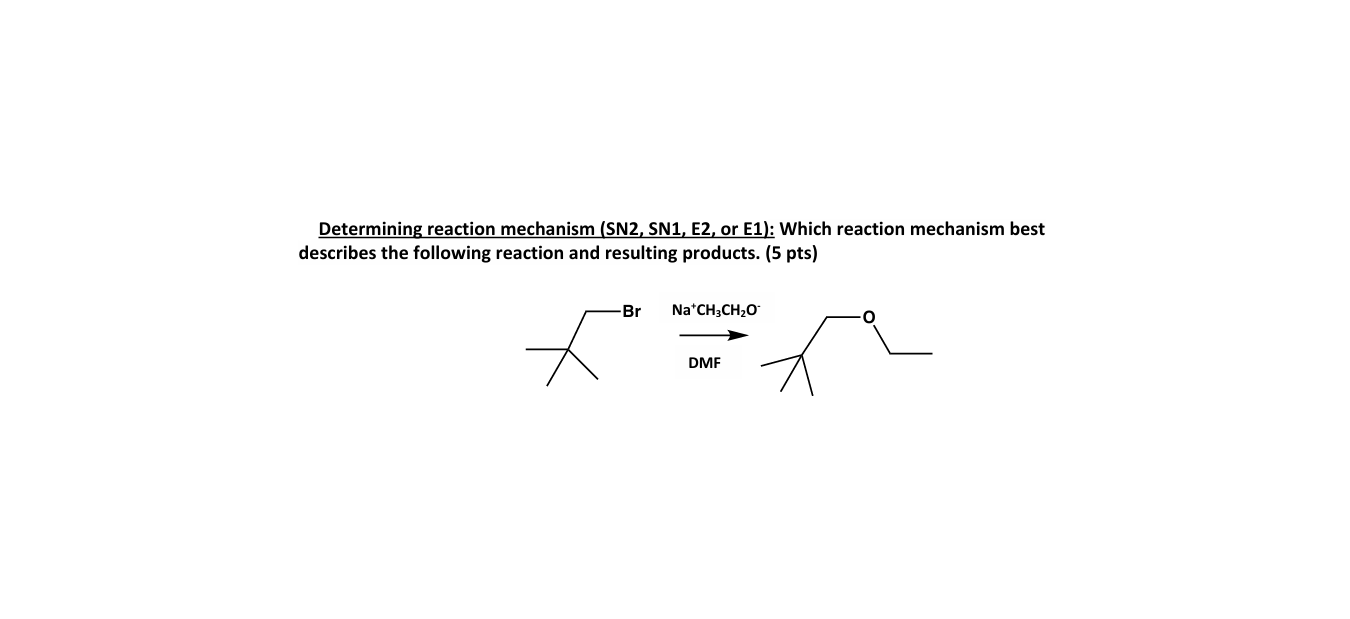
Solved Determining Reaction Mechanism Sn2 ï Sn1 ï E2 ï Or Chegg Keeping this in mind, what we see is that essentially you will never have to choose between s n 1 vs e2 and s n 2 or e1 vs e2 and s n 2. that is, if you know the most common strong and weak bases nucleophiles (see below). the weak reactants are mainly going to be the water and alcohols. A flow chart for deciding between sn1, sn2, e1, and e2 mechanisms is explained. the ultimate decision matrix for discerning which reaction pathway prevails under various conditions!. Sn1 vs sn2 vs e1 vs e2: how to choose the right mechanism. decision flowchart based on substrate, nucleophile, base, and solvent. worked examples for organic chemistry exams. It is relatively easy to separate s n 2 and e2 pathways from s n 1 e1, since both s n 2 and e2 require strong nucleophile or strong base that are usually negatively charged species, while s n 1 e1 require neutral conditions.
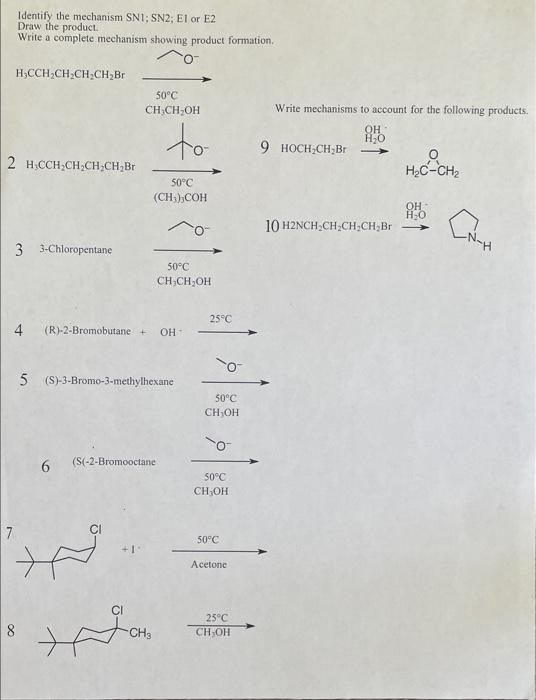
Solved Identify The Mechanism Sn1 Sn2 E1 Or E2 Draw The Chegg Sn1 vs sn2 vs e1 vs e2: how to choose the right mechanism. decision flowchart based on substrate, nucleophile, base, and solvent. worked examples for organic chemistry exams. It is relatively easy to separate s n 2 and e2 pathways from s n 1 e1, since both s n 2 and e2 require strong nucleophile or strong base that are usually negatively charged species, while s n 1 e1 require neutral conditions. Tertiary alkyl halides e2 elimination occurs when a base is used, but s n 1 substitution and e1 elimination occur together under neutral conditions, such as in pure ethanol or water. Comprehensive organic chemistry study guide covering sn1, sn2, e1, and e2 mechanisms, carbocation stability, leaving groups, and zaitsev’s rule. Sn2 and e2 are bimolecular, single step mechanisms involving strong nucleophiles or bases, while sn1 and e1 are unimolecular, multi step mechanisms proceeding through carbocation intermediates and generally involve weak nucleophiles or bases. How to identify where substitution (sn1 and sn2) and elimination reactions (e1 and e2) will plausibly occur by analyzing the leaving group.

Sn2 Reaction Mechanism Tertiary alkyl halides e2 elimination occurs when a base is used, but s n 1 substitution and e1 elimination occur together under neutral conditions, such as in pure ethanol or water. Comprehensive organic chemistry study guide covering sn1, sn2, e1, and e2 mechanisms, carbocation stability, leaving groups, and zaitsev’s rule. Sn2 and e2 are bimolecular, single step mechanisms involving strong nucleophiles or bases, while sn1 and e1 are unimolecular, multi step mechanisms proceeding through carbocation intermediates and generally involve weak nucleophiles or bases. How to identify where substitution (sn1 and sn2) and elimination reactions (e1 and e2) will plausibly occur by analyzing the leaving group.
Comments are closed.