Sn1 Sn2 Mechanism Pptx

Sn1 Mechanism Pdf The document provides an overview of nucleophilic substitution reactions, specifically focusing on sn1 and sn2 mechanisms, their characteristics, and differences. 6. nucleophilic substitution reactions 6.1a: the sn2 mechanism resources for students: ‘guide (summary) comparing sn1 and sn2 reactions and mechanisms’ ‘diagnostic chart for substitution, addition and elimination reactions’ ‘substitution reaction mechanism animations’ sn2: a concerted mechanism.

Sn1 Pptx It discusses the sn1 and sn2 mechanisms, kinetics, stereochemistry, substrate structure, nucleophiles, leaving groups, and solvent effects for sn2 reactions. examples of common and iupac names for alkyl halides are also given. Finally, it discusses factors like temperature, nucleophile strength, and substrate structure that determine whether a reaction will proceed by sn1 or sn2. download as a pptx, pdf or view online for free. The williamson ether synthesis (1850) converts primary alcohols to ethers in two steps. 1. a strong base deprotonates the alcohol. 1. sn2: alkoxide substitutes for bromide. methyl bromide is used to carry the methyl group, acting a bit like sam. these reactants wouldn’t work in a cell or in the body! williamson with 2º or 3º alkyl halides?. Second halogenoalkanes undergo a mixture of the sn1 and sn2 reactions mechanism when reacting with sodium hydroxide. • these are not discussed heavily in sl chemistry.

Sn1 Sn2 Reactions And Their Reaction Mechanisms Pptx The williamson ether synthesis (1850) converts primary alcohols to ethers in two steps. 1. a strong base deprotonates the alcohol. 1. sn2: alkoxide substitutes for bromide. methyl bromide is used to carry the methyl group, acting a bit like sam. these reactants wouldn’t work in a cell or in the body! williamson with 2º or 3º alkyl halides?. Second halogenoalkanes undergo a mixture of the sn1 and sn2 reactions mechanism when reacting with sodium hydroxide. • these are not discussed heavily in sl chemistry. The sn2 reaction sn2 reaction short for substitution, nucleophilic, bimolecular a mechanism that accounts for both the inversion of configuration and the second order kinetics that are observed with nucleophilic substitution reactions suggested in 1937 by e. d. hughes and christopher ingold essential features of the sn2 reaction takes place in. Sn2 reactions the nucleophile: when the attacking atoms are of similar size nucleophilicity parallels basicity. when they are of different size, the larger more polarizable atoms are more nucleophilic the solvent: occur faster in polar aprotic solvents, dmso is a good polar aprotic solvent. Sn2 is a one step, bimolecular reaction where bond breaking and formation occur simultaneously. it favors primary halogenoalkanes due to less steric hindrance allowing frontside attack. A helpful generalization is that compounds that can form relatively stable cations generally do so and react by the sn1 mechanism while the others have to react by the sn2 mechanism.

Sn1 Sn2 Reactions And Their Reaction Mechanisms Pptx The sn2 reaction sn2 reaction short for substitution, nucleophilic, bimolecular a mechanism that accounts for both the inversion of configuration and the second order kinetics that are observed with nucleophilic substitution reactions suggested in 1937 by e. d. hughes and christopher ingold essential features of the sn2 reaction takes place in. Sn2 reactions the nucleophile: when the attacking atoms are of similar size nucleophilicity parallels basicity. when they are of different size, the larger more polarizable atoms are more nucleophilic the solvent: occur faster in polar aprotic solvents, dmso is a good polar aprotic solvent. Sn2 is a one step, bimolecular reaction where bond breaking and formation occur simultaneously. it favors primary halogenoalkanes due to less steric hindrance allowing frontside attack. A helpful generalization is that compounds that can form relatively stable cations generally do so and react by the sn1 mechanism while the others have to react by the sn2 mechanism.
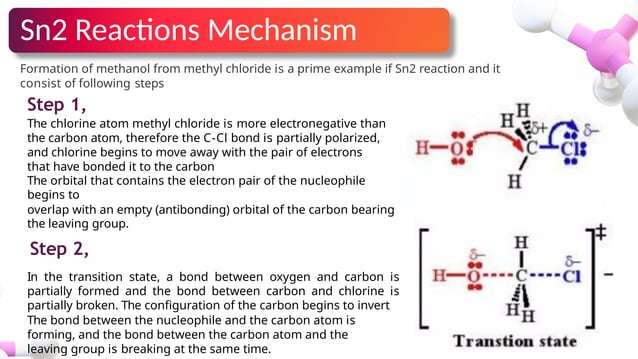
Sn1 Sn2 Reactions And Their Reaction Mechanisms Pptx Sn2 is a one step, bimolecular reaction where bond breaking and formation occur simultaneously. it favors primary halogenoalkanes due to less steric hindrance allowing frontside attack. A helpful generalization is that compounds that can form relatively stable cations generally do so and react by the sn1 mechanism while the others have to react by the sn2 mechanism.

Sn1 Sn2 Reactions And Their Reaction Mechanisms Pptx
Comments are closed.