Sn1 Mechanism Pdf

Sn1 Mechanism Pdf Illustrative reaction: the most common example of an sn1 reaction is the formation of alcohols from alkyl halides as shown below. mechanism involved: the proposed mechanism for the reaction given above involves two steps which must be discussed before we give salient features of the same. The sn1 reaction is a substitution reaction in organic chemistry. "sn" stands for nucleophilic substitution and the " 1" represents the fact that the rate determining step is unimolecular.

Sn1 Mechanism Pdf Chemical Reactions Aromaticity C lg r2 r3 always sn1 (due to stability of tertiary carbocations and steric hindrance of backside attack). Sn2 , sn1 , e2 , & e1: substitution and elimination reactions nucleophilic substitution reactions (sn2 and sn1) replace a leaving group with a nucleophile (nu: or nu: ) elimination reactions (e2 and e1) generate a double bond by loss of " a " and " b: " they may compete with each other. The document discusses the sn1 reaction mechanism, highlighting its distinct characteristics compared to the sn2 mechanism, such as its dependence on substrate concentration and the formation of a carbocation intermediate. All carbocations are very good electrophiles (electron acceptors) and even weak nucleophiles, like h2o and methanol, will react quickly with them. the two sn1 reactions will proceed at essentially the same rate since the only difference is the nucleophile.

Sn1 Mechanism Objectives Pdf The document discusses the sn1 reaction mechanism, highlighting its distinct characteristics compared to the sn2 mechanism, such as its dependence on substrate concentration and the formation of a carbocation intermediate. All carbocations are very good electrophiles (electron acceptors) and even weak nucleophiles, like h2o and methanol, will react quickly with them. the two sn1 reactions will proceed at essentially the same rate since the only difference is the nucleophile. Substitution nucleophilic unimolecular (sn1) the meaning of sn1 reaction lies in the name itself “substitution, nucleophilic, and unimolecular.” therefore, this reaction will follow the first order kinetics. in another term, we can say that the rate determining step in this reaction is unimolecular. The reaction between tert butylbromide and water proceeds via the sn1 mechanism. unlike s n 2 that is a single step reaction, s n 1 reaction involves multiple steps. Sn1 reactions are nucleophilic substitutions that proceed through a carbocation intermediate. they are unimolecular, depending only on the concentration of one reactant, and lose stereochemistry. All carbocations are very good electrophiles (electron acceptors) and even weak nucleophiles, like h2o and methanol, will react quickly with them. the two sn1 reactions will proceed at essentially the same rate since the only difference is the nucleophile.
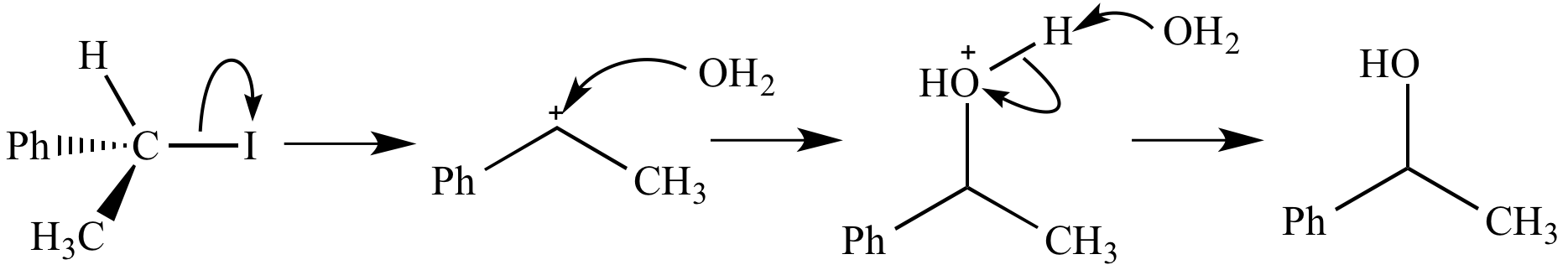
Illustrated Glossary Of Organic Chemistry Sn1 Mechanism Substitution nucleophilic unimolecular (sn1) the meaning of sn1 reaction lies in the name itself “substitution, nucleophilic, and unimolecular.” therefore, this reaction will follow the first order kinetics. in another term, we can say that the rate determining step in this reaction is unimolecular. The reaction between tert butylbromide and water proceeds via the sn1 mechanism. unlike s n 2 that is a single step reaction, s n 1 reaction involves multiple steps. Sn1 reactions are nucleophilic substitutions that proceed through a carbocation intermediate. they are unimolecular, depending only on the concentration of one reactant, and lose stereochemistry. All carbocations are very good electrophiles (electron acceptors) and even weak nucleophiles, like h2o and methanol, will react quickly with them. the two sn1 reactions will proceed at essentially the same rate since the only difference is the nucleophile.
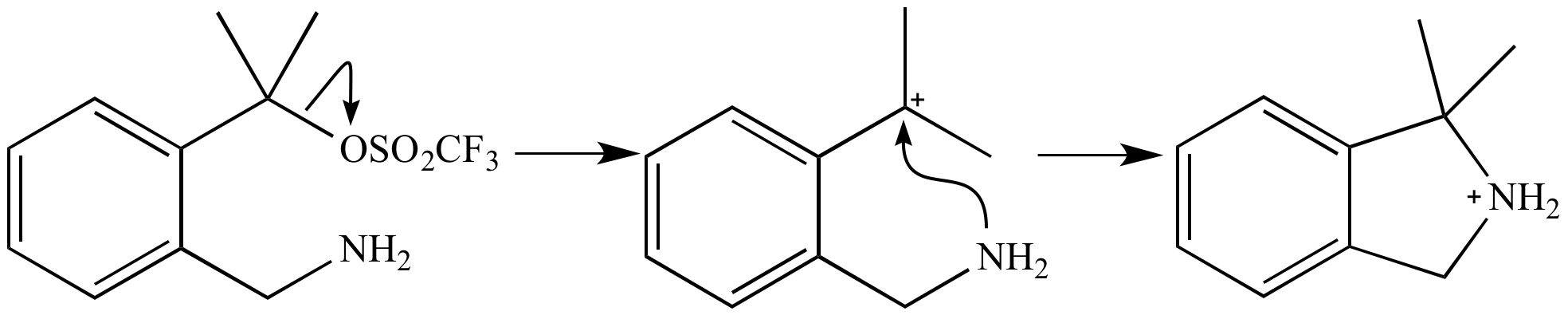
Illustrated Glossary Of Organic Chemistry Sn1 Mechanism Sn1 reactions are nucleophilic substitutions that proceed through a carbocation intermediate. they are unimolecular, depending only on the concentration of one reactant, and lose stereochemistry. All carbocations are very good electrophiles (electron acceptors) and even weak nucleophiles, like h2o and methanol, will react quickly with them. the two sn1 reactions will proceed at essentially the same rate since the only difference is the nucleophile.
Comments are closed.