Process Validation And Re Validation Study
Process Validation Re Validation Report Excel Pdf Sample Process characterization is a set of documented studies in which operational parameters are purposely varied to determine their effect on product quality attributes and process performance. The overarching text presented in this annex constitutes the general principles of the new guidance on validation. the following appendices included in this annex address specific aspects of validation and are intended to complement the general text on validation:.
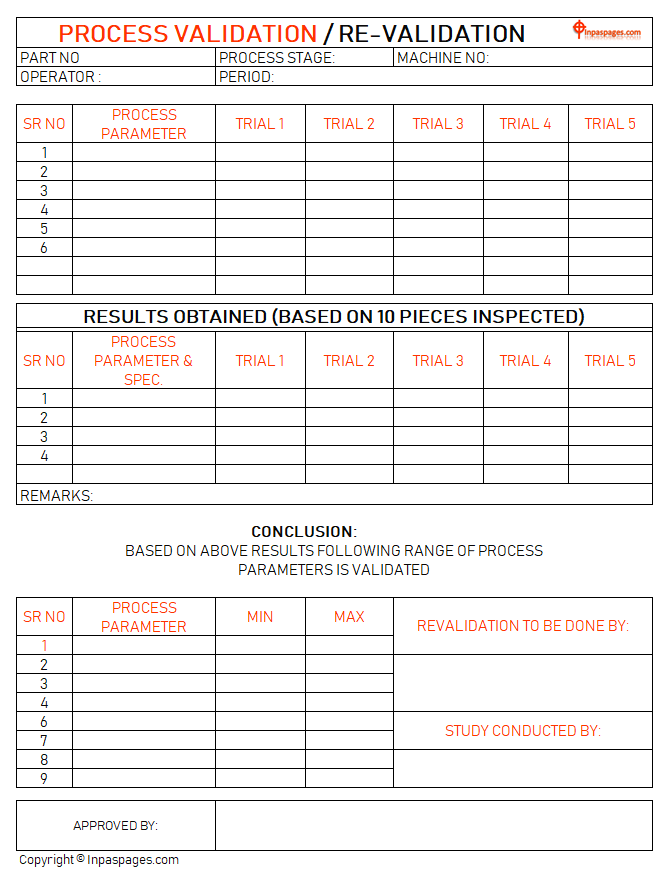
Process Validation And Re Validation Study Actually, process validation is key analytical process for ensuring process reliability and capability of consistent to product quality. these procedure defines the values of criteria based on evaluation of data collected from stage of production. Medical device product quality directly affects the safety of life of patients. therefore, process validation is more important to the medical device manufacture. it is not only for regulations, but also is a way to ensure that the manufacturing process is continuing effective. The guideline is brought into line with ich q8, q9 and q10 documents and the possibility to use continuous process verification in addition to, or instead of, traditional process validation described in the previous guideline has been added and is encouraged. Process validation will ensure a robust product that is highly reproducible over time. major changes after the initial validation will result in the need for subsequent revalidation.

Process Re Validation Presentationeze The guideline is brought into line with ich q8, q9 and q10 documents and the possibility to use continuous process verification in addition to, or instead of, traditional process validation described in the previous guideline has been added and is encouraged. Process validation will ensure a robust product that is highly reproducible over time. major changes after the initial validation will result in the need for subsequent revalidation. The revised process validation (pv) guidance from fda (january 2011) aligns process validation activities with a product lifecycle concept, emphasizing the expectation that process validation starts with process design and spans the entire lifecycle of the marketed product. When an established platform analytical procedure is used for a new purpose, validation testing can be abbreviated, if scientifically justified. approaches other than those set forth in this guideline may be applicable and acceptable with appropriate science based justification. Facilities, processes, and process controls that are currently in use but have not gone through a formally documented validation procedure are validated retrospectively. This phase requires frequent review of all process related documents, including validation audit report to assure that there have been no changes, deviations, failures, modifications to the production process and that all sop have been followed, including change control procedure.
Comments are closed.