Process Re Validation Presentationeze
Process Validation Re Validation Report Excel Pdf Sample Any change within a product or process, including changes to personnel, equipment, procedures, etc., need to be evaluated to determine the potential consequences on validation status. This document discusses process validation, which involves collecting data throughout production to demonstrate a process can consistently deliver quality products. it describes the three stages of process validation: process design, process qualification, and continuous process verification.
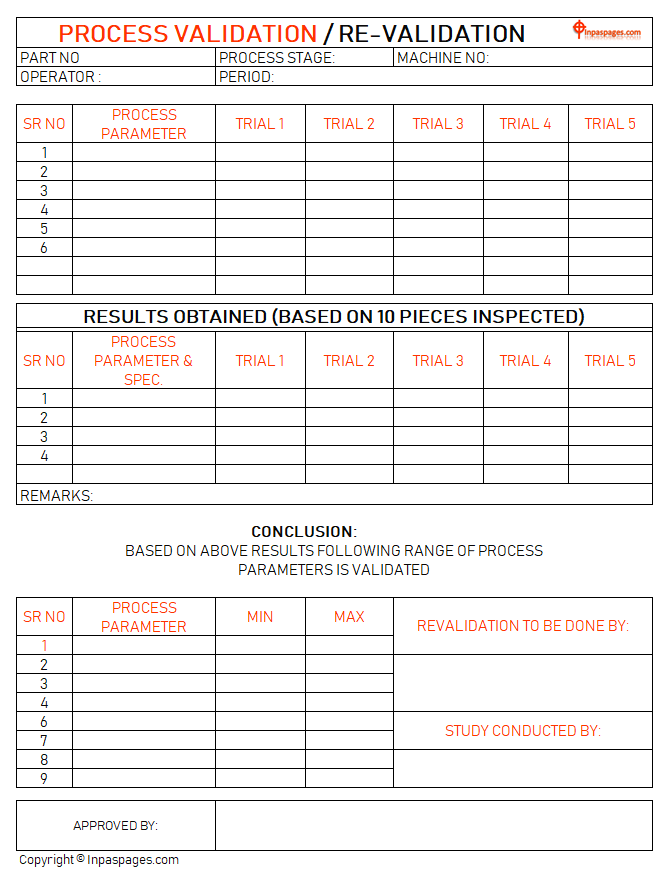
Process Validation And Re Validation Study In planning a product and process validation, a common approach is to create a top level validation master plan, from which the review, approval & implementation process for the various protocols and test activities can be clearly controlled. The following sampling plan and acceptance criteria provide a guide for process validation of a typical solid oral dosage manufacturing process with medium risk indication. Step through at your own pace, focus on topics of interest to you refresh and build upon your current level of knowledge the cost is detailed upfront. there are no extra or hidden costs. This presentation discusses process validation, including its definition, scope, objectives, types (prospective, retrospective, concurrent, revalidation), stages, responsibilities of different departments, protocols, sampling procedures, acceptance criteria, and reports.

Process Re Validation Presentationeze Step through at your own pace, focus on topics of interest to you refresh and build upon your current level of knowledge the cost is detailed upfront. there are no extra or hidden costs. This presentation discusses process validation, including its definition, scope, objectives, types (prospective, retrospective, concurrent, revalidation), stages, responsibilities of different departments, protocols, sampling procedures, acceptance criteria, and reports. This document outlines the process validation stages in pharmaceutical manufacturing, which include process design, process qualification, and continued process verification. The validation lifecycle is an approach to product and process validation from concept stage, through to end customer use, with a continuous feedback loop, that confirms continued adherence from the original design specifications through to end customer requirements. You are here homepage medical device validation medical device validation full details process re validation. The presentation by dr. arti r. thakkar discusses the essentials of process validation in pharmaceutical production, defining it as the documented evidence ensuring that processes consistently produce products meeting predetermined specifications.
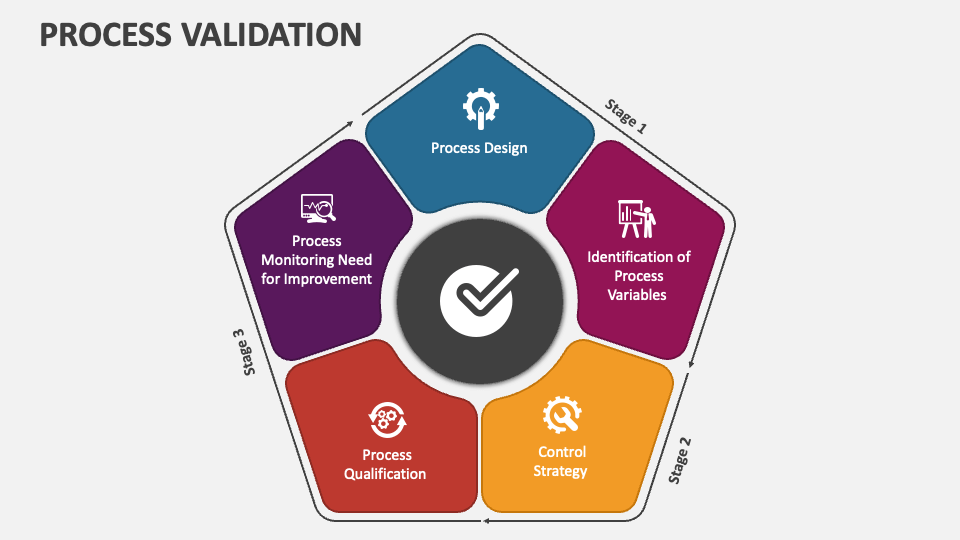
Process Validation Powerpoint Presentation Slides Ppt Template This document outlines the process validation stages in pharmaceutical manufacturing, which include process design, process qualification, and continued process verification. The validation lifecycle is an approach to product and process validation from concept stage, through to end customer use, with a continuous feedback loop, that confirms continued adherence from the original design specifications through to end customer requirements. You are here homepage medical device validation medical device validation full details process re validation. The presentation by dr. arti r. thakkar discusses the essentials of process validation in pharmaceutical production, defining it as the documented evidence ensuring that processes consistently produce products meeting predetermined specifications.
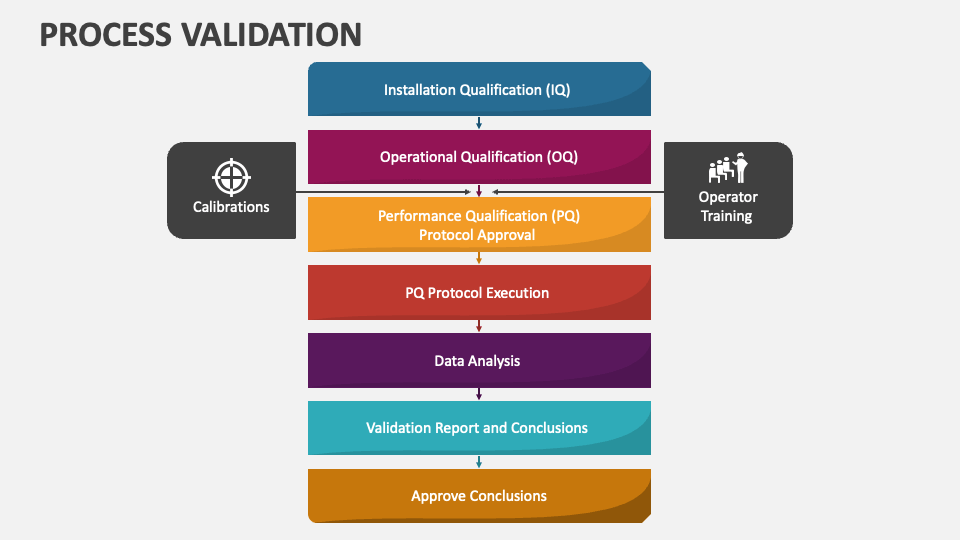
Process Validation Powerpoint Presentation Slides Ppt Template You are here homepage medical device validation medical device validation full details process re validation. The presentation by dr. arti r. thakkar discusses the essentials of process validation in pharmaceutical production, defining it as the documented evidence ensuring that processes consistently produce products meeting predetermined specifications.
Comments are closed.