Periodic Table Trends Electron Affinity

Periodic Table Trends Electron Affinity Period and group trends for electron affinities are not nearly as regular as for ionization energy. in general, electron affinities increase (become more negative) from left to right across a period and decrease (become less negative) from top to bottom down a group. Learn about electron affinity in chemistry. see the electron affinity trend and learn about the elements with the lowest and highest values.
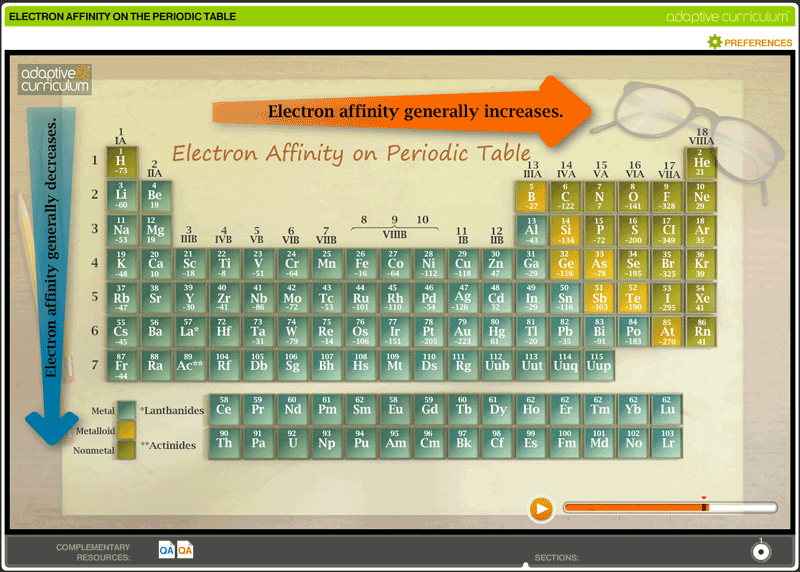
Electron Affinity Trends Of The Periodic Table Below is a visual representation of electron affinity trends throughout the periodic table. as discussed, electron affinities increase from left to right across periods; electron affinities decrease from top to bottom down groups. What is electron affinity. learn its equation, examples, values, and change across a period & down a group in the periodic table. also, learn its exceptions. This easy to use chart shows the periodic table trends of electronegativity, ionization energy, atomic radius, metallic character, and electron affinity. Explore how electron affinity changes with atomic number in the periodic table of elements via interactive plots.
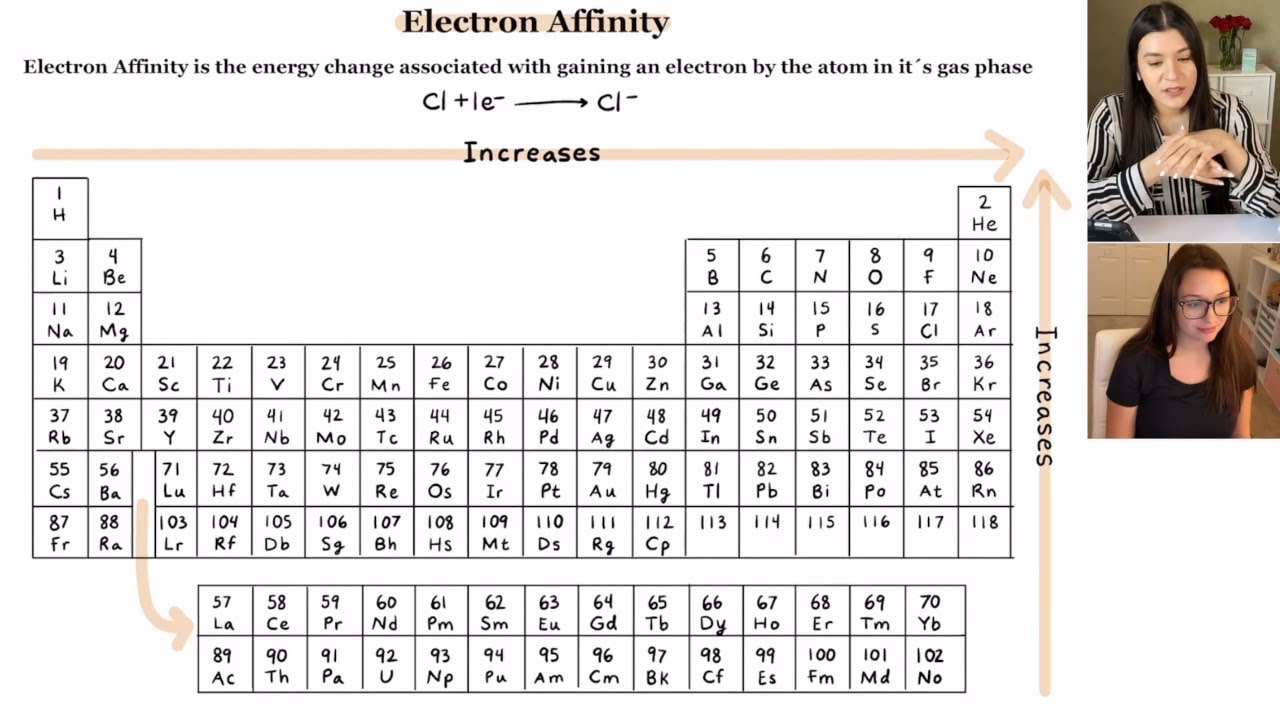
Periodic Table Electron Affinity Webelements Periodic Table This easy to use chart shows the periodic table trends of electronegativity, ionization energy, atomic radius, metallic character, and electron affinity. Explore how electron affinity changes with atomic number in the periodic table of elements via interactive plots. Understanding electron affinity trends across the periodic table reveals why certain elements readily form anions while others resist gaining electrons entirely. Learn what electron affinity means, its trends across the periodic table, real examples, exceptions, and how it differs from electronegativity for exam success. Learn the forces—nuclear charge and atomic size—that drive electron affinity trends across the periodic table, including key exceptions. Periodic table trends explained: atomic radius, ionization energy, and electron affinity — why they trend, how to predict, and the exceptions tested on chemistry exams.

4 Electron Affinity Periodic Table Trend Images Stock Photos Vectors Understanding electron affinity trends across the periodic table reveals why certain elements readily form anions while others resist gaining electrons entirely. Learn what electron affinity means, its trends across the periodic table, real examples, exceptions, and how it differs from electronegativity for exam success. Learn the forces—nuclear charge and atomic size—that drive electron affinity trends across the periodic table, including key exceptions. Periodic table trends explained: atomic radius, ionization energy, and electron affinity — why they trend, how to predict, and the exceptions tested on chemistry exams.
Comments are closed.