Pathways To U S Fda Drug Approval

Fda Approval Pathways Devices Get to know fda’s drug development and approval process ensuring that drugs work and that the benefits outweigh their known risks. This article provides a comprehensive tutorial on the primary fda submission pathways: investigational new drug (ind), new drug application (nda), abbreviated new drug application (anda), biologics license application (bla), and the 505 (b) (2) application.

Fda Approval Pathways Devices This guide explains how the fda approval process works from start to finish — the submissions, review types, decision points, and pathways that shape drug development strategy and create the catalysts that move markets. The drug approval by fda is a lengthy process but it is helpful for pharmaceutical regulation and public health protection. the whole process is detailed and demanding from pre clinical research to post market surveillance. Every stage of fda drug approval explained — from preclinical research through phase 1 3 clinical trials to nda submission and post market surveillance. Ask the main expedited regulatory pathways for the development and approval of prescription drugs by the us food and drug administration (fda) are priority review, fast track, accelerated approval, and breakthrough therapy.

Fda Approval Pathways For Aging Every stage of fda drug approval explained — from preclinical research through phase 1 3 clinical trials to nda submission and post market surveillance. Ask the main expedited regulatory pathways for the development and approval of prescription drugs by the us food and drug administration (fda) are priority review, fast track, accelerated approval, and breakthrough therapy. Learn how the fda drug approval process works, including key steps, timelines, clinical trials, and what to expect before a drug reaches patients. Understand the full fda drug approval process from preclinical research to post marketing surveillance. learn how drugs are tested, reviewed, and monitored in the u.s. There are multiple routes for approval of new drugs or indications for existing medications through the united states food and drug administration (fda), many of which are targeted for therapies in niche or rare disease treatment areas. Requires the fda to provide a summary of the basis of approval for a drug approved through the accelerated approval pathway, including whether an advisory committee meeting was held, and the rationale for determining the surrogate endpoint is reasonably likely to predict clinical benefit.

Fda Approval Pathways For Aging Learn how the fda drug approval process works, including key steps, timelines, clinical trials, and what to expect before a drug reaches patients. Understand the full fda drug approval process from preclinical research to post marketing surveillance. learn how drugs are tested, reviewed, and monitored in the u.s. There are multiple routes for approval of new drugs or indications for existing medications through the united states food and drug administration (fda), many of which are targeted for therapies in niche or rare disease treatment areas. Requires the fda to provide a summary of the basis of approval for a drug approved through the accelerated approval pathway, including whether an advisory committee meeting was held, and the rationale for determining the surrogate endpoint is reasonably likely to predict clinical benefit.
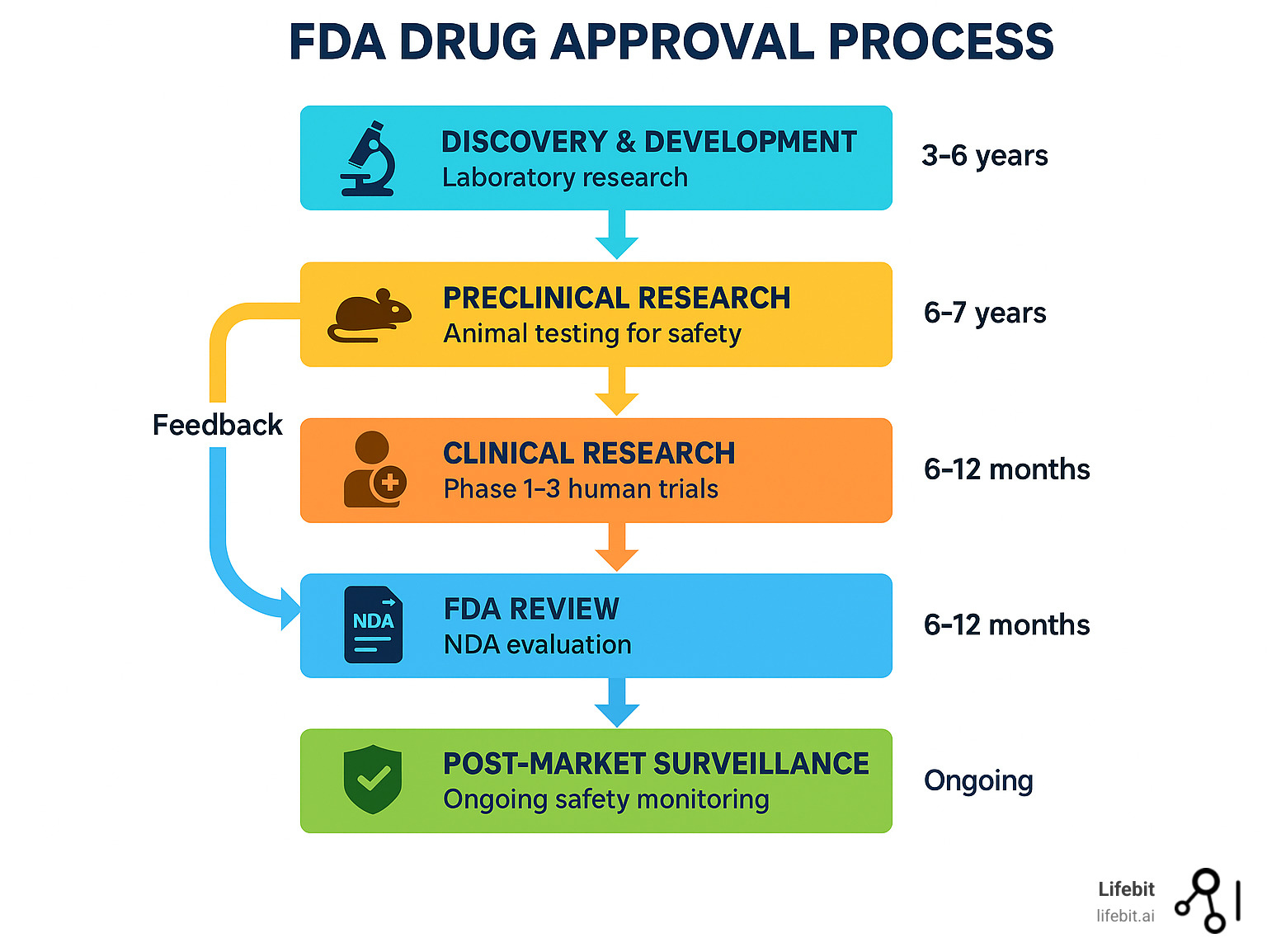
Fda Drug Approval Process 5 Easy Steps There are multiple routes for approval of new drugs or indications for existing medications through the united states food and drug administration (fda), many of which are targeted for therapies in niche or rare disease treatment areas. Requires the fda to provide a summary of the basis of approval for a drug approved through the accelerated approval pathway, including whether an advisory committee meeting was held, and the rationale for determining the surrogate endpoint is reasonably likely to predict clinical benefit.
Comments are closed.