Fda Drug Approval Process 5 Easy Steps

Guide To The Fda Drug Approval Process Pdf Understand the fda drug approval process. our guide simplifies every step, from trials to market, ensuring drug safety and efficacy. Get to know fda’s drug development and approval process ensuring that drugs work and that the benefits outweigh their known risks.

Fda Drug Approval Process Learn how the fda drug approval process works, including key steps, timelines, clinical trials, and what to expect before a drug reaches patients. Complete fda drug approval process step by step. preclinical testing, ind, phase 1 3 trials, nda bla review, advisory committee, inspections, and approval. The fda approval process for new medications involves five core stages: discovery and development, preclinical research, clinical research, fda review, and post market safety monitoring. Drug approval by the fda is a critical process and has to follow different steps before any drug product reaches to the market. the following steps are followed by any company to get approval from the fda to produce and market any drug product.

Fda Drug Approval Process Steps At Maggie Martinez Blog The fda approval process for new medications involves five core stages: discovery and development, preclinical research, clinical research, fda review, and post market safety monitoring. Drug approval by the fda is a critical process and has to follow different steps before any drug product reaches to the market. the following steps are followed by any company to get approval from the fda to produce and market any drug product. This guide explains how the fda approval process works from start to finish — the submissions, review types, decision points, and pathways that shape drug development strategy and create the catalysts that move markets. Learn how a new drug gets fda approval step by step, from clinical trials to nda review and final authorization. Understand the full fda drug approval process from preclinical research to post marketing surveillance. learn how drugs are tested, reviewed, and monitored in the u.s. In this guide, we’ll break down the drug development process step by step, including timelines, success rates, regulatory pathways, and the operational challenges pharmaceutical companies face along the way.
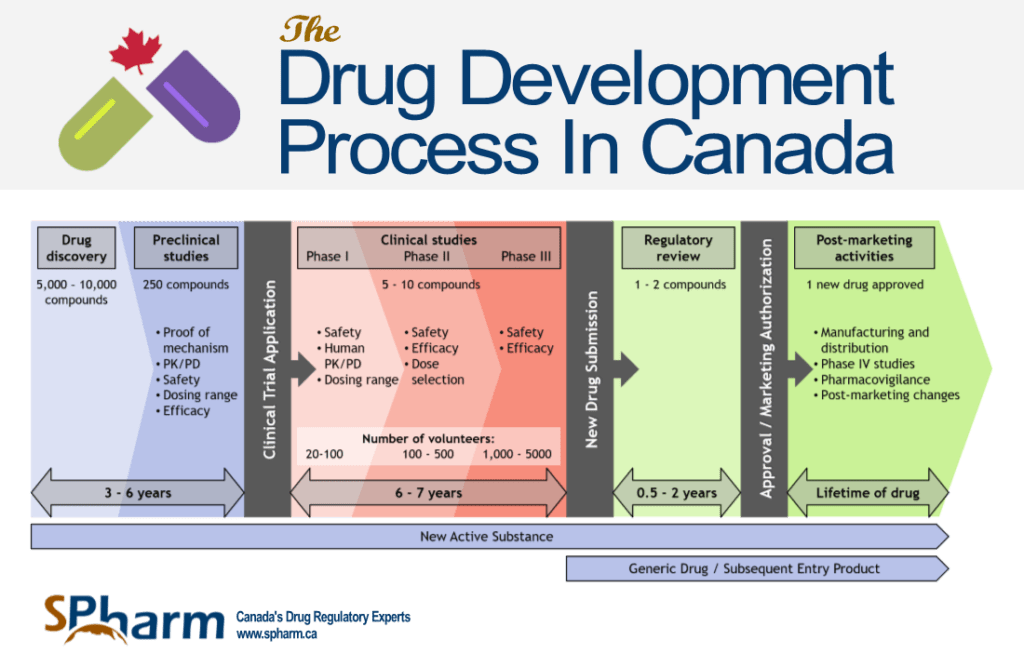
Fda Drug Approval Process Steps At Maggie Martinez Blog This guide explains how the fda approval process works from start to finish — the submissions, review types, decision points, and pathways that shape drug development strategy and create the catalysts that move markets. Learn how a new drug gets fda approval step by step, from clinical trials to nda review and final authorization. Understand the full fda drug approval process from preclinical research to post marketing surveillance. learn how drugs are tested, reviewed, and monitored in the u.s. In this guide, we’ll break down the drug development process step by step, including timelines, success rates, regulatory pathways, and the operational challenges pharmaceutical companies face along the way.
Comments are closed.