Oxidation Reduction Concept Pdf

Oxidation Reduction Concept Pdf In this chapter we will be introducing the chemistry of reduction oxidation (redox) reactions. this important reaction class is defined by changes in oxidation states for one or more reactant elements, and it includes a subset of reactions involving the transfer of electrons between reactant species. They also use the term oxidation to describe many similar reactions that do not have oxygen as a reactant. this section explains the meaning of oxidation and shows why oxidation is coupled with a corresponding chemical change called reduction.

Oxidation Reduction Pdf Layers Of Learning S a major stumbling block for students. remember oxidation is the loss of electrons and reduction is the gain of electrons. a reducing agent is an electron donor (it loses electrons and is oxidized), and an oxidizing agent is an electron accep or (it gains electrons and is reduced). in terms of our example, zn cu 2 zn 2 cu table 4–1. The reaction involving the transfer of electrons from one molecule to another are called oxidation reduction reactions or redox reactions. these reactions are used in the synthesis of a large variety of compounds and are known in biological systems as well. Oxidation reduction reactions, or redox reactions, are technically defined as any chemical reaction in which the oxidation number of the participating atom, ion, or molecule of a chemical compound changes. In ionic and free radical reactions, oxidation and reduction are defined as processes by which an element undergoes a net loss or gain of electrons, respectively.

Oxidation Reduction Oxidation reduction reactions, or redox reactions, are technically defined as any chemical reaction in which the oxidation number of the participating atom, ion, or molecule of a chemical compound changes. In ionic and free radical reactions, oxidation and reduction are defined as processes by which an element undergoes a net loss or gain of electrons, respectively. Chapter 8 of 'chemistry for changing times' focuses on oxidation and reduction (redox) reactions, explaining the concepts of oxidation as the loss of electrons and reduction as the gain of electrons. Classical concept of oxidation reduction. oxidation : according to this concept, oxidation is considered as the addition of oxygen or removal of hydrogen in an ion, in a compound or in a species. Rtance in all living systems. photosynthesis generates compounds that are powerful reducing agents, and from them electrons are transferred through a series of compounds ultimately to oxygen in either. Using the oxidation number concept, an all inclusive definition of redox reaction has been established. oxidation reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number.

Oxidation Reduction Reactions Nebraska Scientific Chapter 8 of 'chemistry for changing times' focuses on oxidation and reduction (redox) reactions, explaining the concepts of oxidation as the loss of electrons and reduction as the gain of electrons. Classical concept of oxidation reduction. oxidation : according to this concept, oxidation is considered as the addition of oxygen or removal of hydrogen in an ion, in a compound or in a species. Rtance in all living systems. photosynthesis generates compounds that are powerful reducing agents, and from them electrons are transferred through a series of compounds ultimately to oxygen in either. Using the oxidation number concept, an all inclusive definition of redox reaction has been established. oxidation reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number.
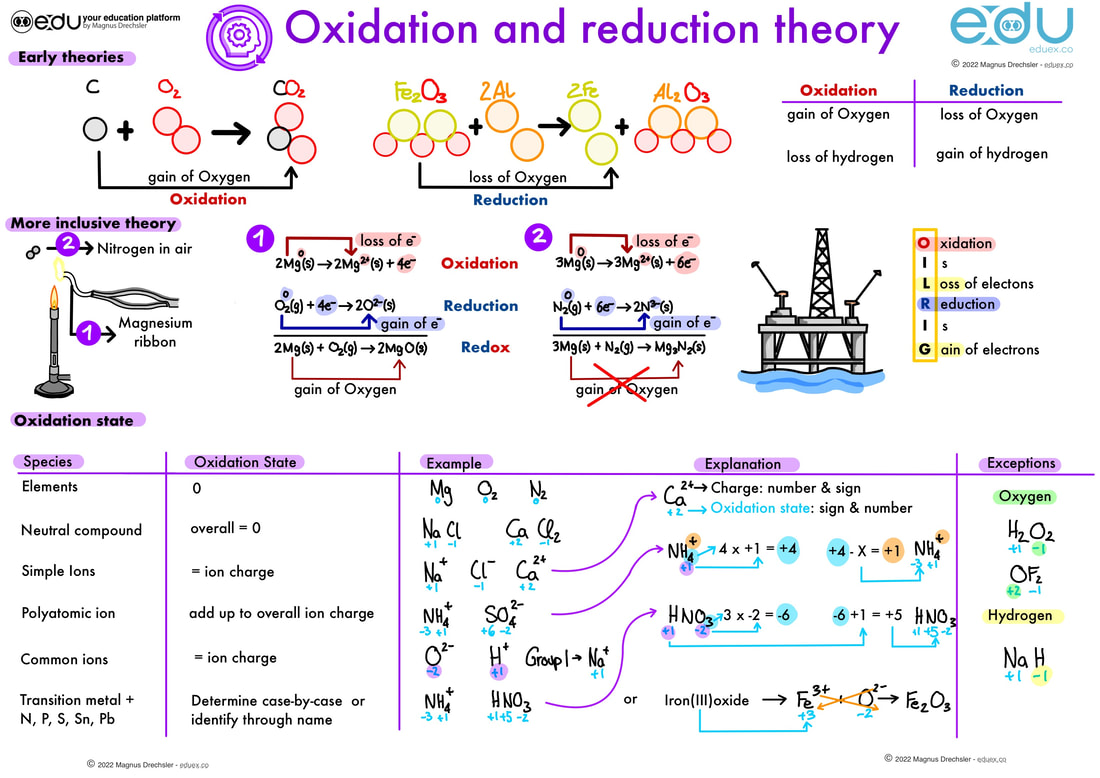
Oxidation And Reduction Eduex Rtance in all living systems. photosynthesis generates compounds that are powerful reducing agents, and from them electrons are transferred through a series of compounds ultimately to oxygen in either. Using the oxidation number concept, an all inclusive definition of redox reaction has been established. oxidation reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number.
Comments are closed.