Oxidation And Reduction Eduex
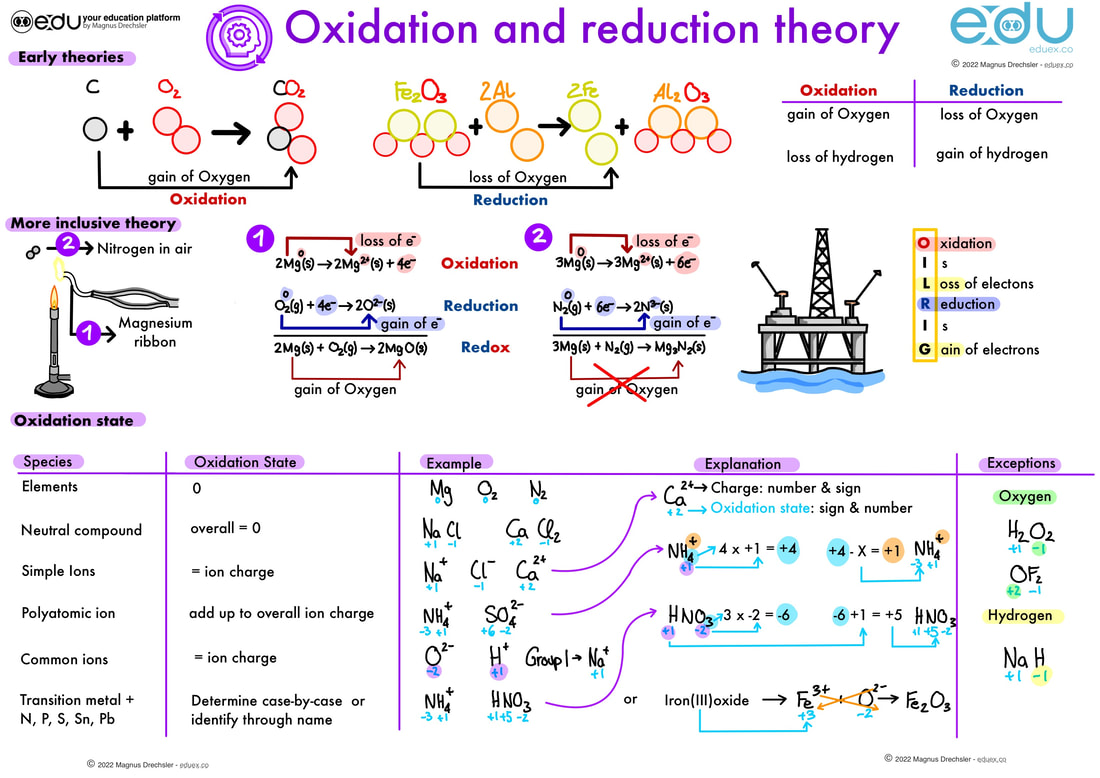
Oxidation And Reduction Eduex This video is introducing the basics of oxidation and reduction as well as explains what oxidation numbers are and how they are useful in redox reactions. task: fill a burette with the potassium iodate solution, kio3 (aq), and measure 5.00 cm3 into a conical flask. This page discusses the various definitions of oxidation and reduction (redox) in terms of the transfer of oxygen, hydrogen, and electrons. it also explains the terms oxidizing agent and reducing ….

Oxidation And Reduction Eduex This tutorial covers oxidation reduction (redox) reactions in electrochemistry: learn to write simple half reactions and to recognize the oxidizing agent and reducing agent. Oxidation reduction reactions (redox reactions) are defined as chemical processes in which electrons are transferred between particles, resulting in changes in their oxidation states. An oxidation reduction reaction is any chemical reaction in which the oxidation number of a participating chemical species changes. many such reactions are common and familiar—combustion, rusting, cellular respiration, and photosynthesis are some examples. Oxidation–reduction (redox) reactions (article) | khan academy khan academy.

Oxidation And Reduction Eduex An oxidation reduction reaction is any chemical reaction in which the oxidation number of a participating chemical species changes. many such reactions are common and familiar—combustion, rusting, cellular respiration, and photosynthesis are some examples. Oxidation–reduction (redox) reactions (article) | khan academy khan academy. Oxidation reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number. returning to the reactions used to introduce this topic, they may now both be identified as redox processes. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. the oxidation and reduction processes occur simultaneously in the chemical reaction. Oxidation–reduction reactions are balanced by separating the overall chemical equation into an oxidation equation and a reduction equation. in oxidation–reduction reactions, electrons are transferred from one substance or atom to another. The term oxidation was originally used to describe chemical reactions involving o 2, but its meaning has evolved to refer to a broad and important reaction class known as oxidation reduction (redox) reactions.
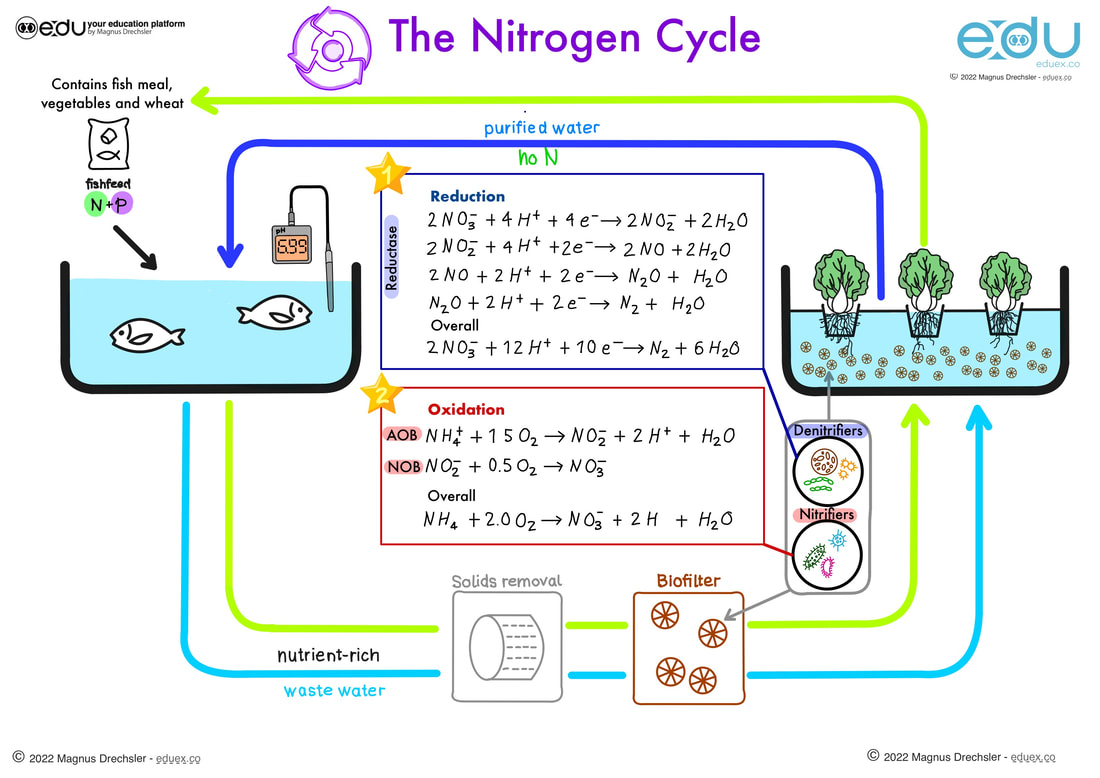
Oxidation And Reduction Eduex Oxidation reduction (redox) reactions are those in which one or more elements involved undergo a change in oxidation number. returning to the reactions used to introduce this topic, they may now both be identified as redox processes. Oxidation is the loss of electrons or an increase in the oxidation state, while reduction is the gain of electrons or a decrease in the oxidation state. the oxidation and reduction processes occur simultaneously in the chemical reaction. Oxidation–reduction reactions are balanced by separating the overall chemical equation into an oxidation equation and a reduction equation. in oxidation–reduction reactions, electrons are transferred from one substance or atom to another. The term oxidation was originally used to describe chemical reactions involving o 2, but its meaning has evolved to refer to a broad and important reaction class known as oxidation reduction (redox) reactions.

9 Oxidation And Reduction Eduex Oxidation–reduction reactions are balanced by separating the overall chemical equation into an oxidation equation and a reduction equation. in oxidation–reduction reactions, electrons are transferred from one substance or atom to another. The term oxidation was originally used to describe chemical reactions involving o 2, but its meaning has evolved to refer to a broad and important reaction class known as oxidation reduction (redox) reactions.

9 Oxidation And Reduction Eduex
Comments are closed.