Limiting Reactants Examples

Limiting Reactants Examples Explain what is meant by the term limiting reagent. how to know which reactant is limiting reagent & how to calculate it. check out a few examples & problems. Now consider a chemical example of a limiting reactant: the production of pure titanium. this metal is fairly light (45% lighter than steel and only 60% heavier than aluminum) and has great mechanical strength (as strong as steel and twice as strong as aluminum).

Limiting Reactants Examples Learn how to solve limiting reactant problems in stoichiometry by identifying which reactant limits the reaction and directly determines the product amount. this lesson offers clear methods, worked out examples, and guidance for confident problem solving. Learn about what a limiting reactant is, how to find a limiting reagent, and see worked out example problems on the limiting reagents. Limiting reactants (higher tier) a reaction finishes when one of the reactants is all used up. the other reactant has nothing left to react with, so some of it is left over:. Use worked examples to identify limiting reactants in chemical reactions. learn more.
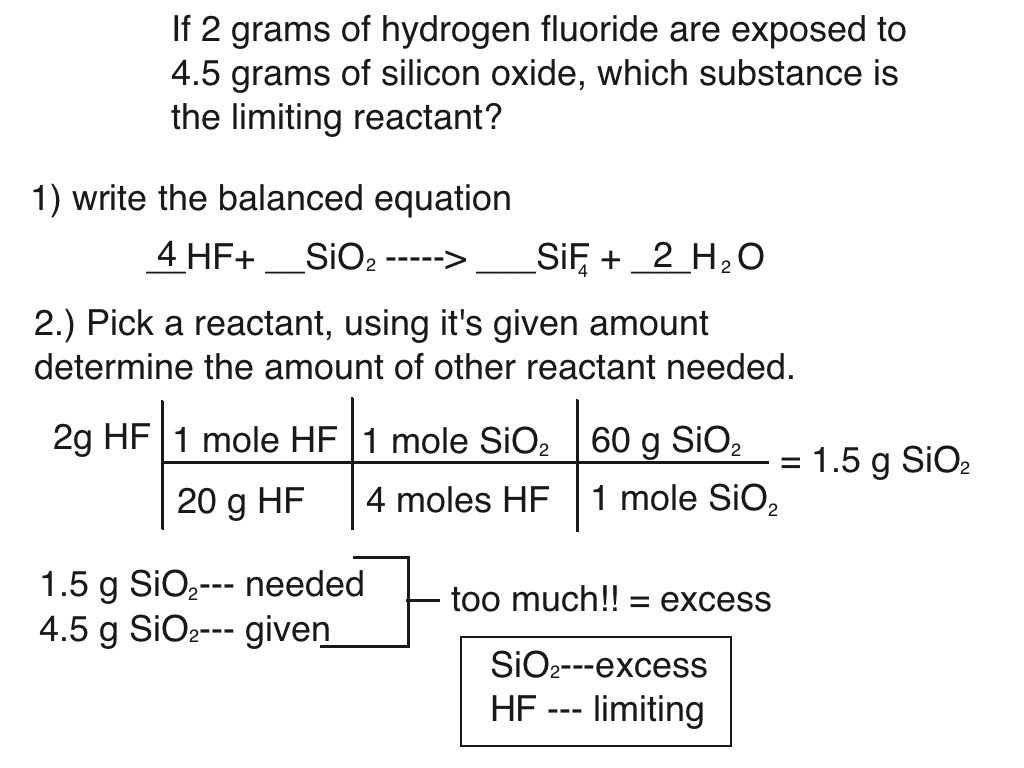
Limiting Reactants Chemistry 101 Limiting reactants (higher tier) a reaction finishes when one of the reactants is all used up. the other reactant has nothing left to react with, so some of it is left over:. Use worked examples to identify limiting reactants in chemical reactions. learn more. Throughout this section, we will delve into the steps for identifying limiting reactants, explore various examples, and understand its application in calculating percent yields. The following stoichiometry road map gives a summary of how to use stoichiometry to calculate moles, masses, volumes and particles in a chemical reaction with limiting and excess reactants. Learn how to identify the limiting reactant in chemical reactions with easy to follow steps. master stoichiometry, mole ratios, and theoretical yield calculations with practice problems and expert explanations. For example, imagine combining 3 moles of h 2 and 2 moles of cl 2. this represents a 3:2 (or 1.5:1) ratio of hydrogen to chlorine present for reaction, which is greater than the stoichiometric ratio of 1:1. hydrogen, therefore, is present in excess, and chlorine is the limiting reactant.

Chemistry Limiting Reactants Practice By Teach Simple Throughout this section, we will delve into the steps for identifying limiting reactants, explore various examples, and understand its application in calculating percent yields. The following stoichiometry road map gives a summary of how to use stoichiometry to calculate moles, masses, volumes and particles in a chemical reaction with limiting and excess reactants. Learn how to identify the limiting reactant in chemical reactions with easy to follow steps. master stoichiometry, mole ratios, and theoretical yield calculations with practice problems and expert explanations. For example, imagine combining 3 moles of h 2 and 2 moles of cl 2. this represents a 3:2 (or 1.5:1) ratio of hydrogen to chlorine present for reaction, which is greater than the stoichiometric ratio of 1:1. hydrogen, therefore, is present in excess, and chlorine is the limiting reactant.
Comments are closed.