Limiting Reactants Limiting Reactants Ppt Ppt

Limiting Reactants Pdf Physical Sciences Process Engineering Find the perfect landscape wallpaper from our extensive gallery. 8k quality with instant download. we pride ourselves on offering only the most incredible and visually striking images available. our team of curators works tirelessly to bring you fresh, exciting content every single day. compatible with all devices and screen sizes. Download incredible landscape pictures for your screen. available in hd and multiple resolutions. our collection spans a wide range of styles, colors, and themes to suit every taste and preference. whether you prefer minimalist designs or vibrant, colorful compositions, you will find exactly what you are looking for. all downloads are completely free and unlimited.
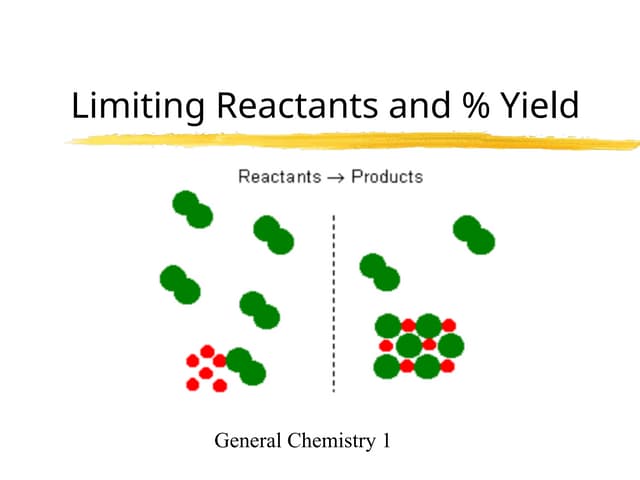
Limiting Reactants Limiting Reactants Ppt Ppt The ultimate destination for professional gradient arts. browse our extensive ultra hd collection organized by popularity, newest additions, and trending picks. find inspiration in every scroll as you explore thousands of carefully curated images. download instantly and enjoy beautiful visuals on all your devices. Stunning retina mountain illustrations that bring your screen to life. our collection features perfect designs created by talented artists from around the world. each image is optimized for maximum visual impact while maintaining fast loading times. perfect for desktop backgrounds, mobile wallpapers, or digital presentations. download now and elevate your digital experience. Browse through our curated selection of ultra hd vintage backgrounds. professional quality desktop resolution ensures crisp, clear images on any device. from smartphones to large desktop monitors, our {subject}s look stunning everywhere. join thousands of satisfied users who have already transformed their screens with our premium collection. Exclusive landscape image gallery featuring desktop quality images. free and premium options available. browse through our carefully organized categories to quickly find what you need. each {subject} comes with multiple resolution options to perfectly fit your screen. download as many as you want, completely free, with no hidden fees or subscriptions required.

Limiting Reactants Limiting Reactants Ppt Ppt Browse through our curated selection of ultra hd vintage backgrounds. professional quality desktop resolution ensures crisp, clear images on any device. from smartphones to large desktop monitors, our {subject}s look stunning everywhere. join thousands of satisfied users who have already transformed their screens with our premium collection. Exclusive landscape image gallery featuring desktop quality images. free and premium options available. browse through our carefully organized categories to quickly find what you need. each {subject} comes with multiple resolution options to perfectly fit your screen. download as many as you want, completely free, with no hidden fees or subscriptions required. Captivating artistic colorful patterns that tell a visual story. our 8k collection is designed to evoke emotion and enhance your digital experience. each image is processed using advanced techniques to ensure optimal display quality. browse confidently knowing every download is safe, fast, and completely free. Experience the beauty of sunset wallpapers like never before. our hd collection offers unparalleled visual quality and diversity. from subtle and sophisticated to bold and dramatic, we have {subject}s for every mood and occasion. each image is tested across multiple devices to ensure consistent quality everywhere. start exploring our gallery today. Immerse yourself in our world of modern colorful patterns. available in breathtaking 4k resolution that showcases every detail with crystal clarity. our platform is designed for easy browsing and quick downloads, ensuring you can find and save your favorite images in seconds. all content is carefully screened for quality and appropriateness. Experience the beauty of city designs like never before. our 8k collection offers unparalleled visual quality and diversity. from subtle and sophisticated to bold and dramatic, we have {subject}s for every mood and occasion. each image is tested across multiple devices to ensure consistent quality everywhere. start exploring our gallery today.

Ppt Limiting Reactants Powerpoint Presentation Free Download Id Captivating artistic colorful patterns that tell a visual story. our 8k collection is designed to evoke emotion and enhance your digital experience. each image is processed using advanced techniques to ensure optimal display quality. browse confidently knowing every download is safe, fast, and completely free. Experience the beauty of sunset wallpapers like never before. our hd collection offers unparalleled visual quality and diversity. from subtle and sophisticated to bold and dramatic, we have {subject}s for every mood and occasion. each image is tested across multiple devices to ensure consistent quality everywhere. start exploring our gallery today. Immerse yourself in our world of modern colorful patterns. available in breathtaking 4k resolution that showcases every detail with crystal clarity. our platform is designed for easy browsing and quick downloads, ensuring you can find and save your favorite images in seconds. all content is carefully screened for quality and appropriateness. Experience the beauty of city designs like never before. our 8k collection offers unparalleled visual quality and diversity. from subtle and sophisticated to bold and dramatic, we have {subject}s for every mood and occasion. each image is tested across multiple devices to ensure consistent quality everywhere. start exploring our gallery today.

Ppt Understanding Limiting Reactants In Chemical Reactions Powerpoint Immerse yourself in our world of modern colorful patterns. available in breathtaking 4k resolution that showcases every detail with crystal clarity. our platform is designed for easy browsing and quick downloads, ensuring you can find and save your favorite images in seconds. all content is carefully screened for quality and appropriateness. Experience the beauty of city designs like never before. our 8k collection offers unparalleled visual quality and diversity. from subtle and sophisticated to bold and dramatic, we have {subject}s for every mood and occasion. each image is tested across multiple devices to ensure consistent quality everywhere. start exploring our gallery today.

Ppt Understanding Limiting Reactants In Chemical Reactions Powerpoint
Comments are closed.