Irb Reviewers Guide

Basic Institutional Review Board Irb Regulations And Review Process An irb staff member must submit the decision on behalf of the committee. if you are a reviewer of the study, record your comments and attach relevant files (such as reviewer checklists) as described in preparing comments for a meeting. Find important documents if you are an irb committee member or reviewer, you may want to access meeting agendas, worksheets, or checklists.

Types Of Review Human Research Protection Program Hrpp When writing your irb protocol, you should answer all questions on the tc irb application template and use the tc reviewer questions as a guide for what irb reviewers will look for in your protocol. Worksheets are guidance materials used by irb reviewers and designated reviewers, for human subjects research in initial reviews, continuing reviews, and modification reviews, to enhance compliance with federal, state, and local requirements. Institutional review board (irb) written procedures:guidance for institutions and irbs. this guidance represents the office for human research protections’ (ohrp’s) and the food and drug administration’s (fda’s) current thinking on this topic. Irbs have origins in the nuremberg code, a set of principles governing research with human subjects developed in the aftermath of world war ii. over the years, the irb system has evolved to become a vital part of ensuring the ethical conduct of research.

Institutional Review Board Irb Division Of Research Institutional review board (irb) written procedures:guidance for institutions and irbs. this guidance represents the office for human research protections’ (ohrp’s) and the food and drug administration’s (fda’s) current thinking on this topic. Irbs have origins in the nuremberg code, a set of principles governing research with human subjects developed in the aftermath of world war ii. over the years, the irb system has evolved to become a vital part of ensuring the ethical conduct of research. Advarra’s sponsor’s guide to irb approval explains when approval is required, how the process works step by step, and how sponsors can streamline timelines while safeguarding research participants. Keep in mind that the basic job of the irb is to protect the rights and welfare of human research participants and facilitate research by using the basic ethical principles from the belmont report. this section summarizes some of the key federal regulations for establishing an irb. To provide guidance on the appropriate content of written procedures, while considering these variations, this guidance provides a written procedures checklist to assist institutions and irbs. Begin the review process of a protocol at the meeting by making the presentation to the irb, using the primary reviewer’s worksheet as a guide, or the amendment or continuing review checklists if appropriate.
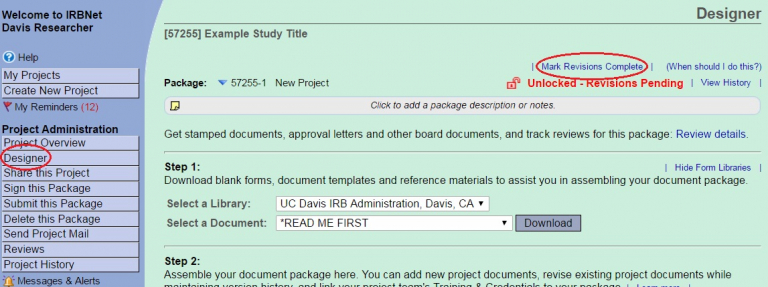
Review Process Irb Advarra’s sponsor’s guide to irb approval explains when approval is required, how the process works step by step, and how sponsors can streamline timelines while safeguarding research participants. Keep in mind that the basic job of the irb is to protect the rights and welfare of human research participants and facilitate research by using the basic ethical principles from the belmont report. this section summarizes some of the key federal regulations for establishing an irb. To provide guidance on the appropriate content of written procedures, while considering these variations, this guidance provides a written procedures checklist to assist institutions and irbs. Begin the review process of a protocol at the meeting by making the presentation to the irb, using the primary reviewer’s worksheet as a guide, or the amendment or continuing review checklists if appropriate.
Comments are closed.