Iconic Bonding Pdf Ion Ionic Bonding

Iconic Bonding Pdf Ion Ionic Bonding An ionic compound is a chemical compound composed of positively charged ions (cations) and negatively charged ions (anions) held together by electrostatic forces (ionic bonds). Iconic bonding free download as pdf file (.pdf), text file (.txt) or read online for free. this document provides an overview and learning objectives for four lessons on ionic and covalent bonding.

Ionic Bonding Pdf Ionic Bonding Ion Ionic and covalent bonds are both strong, but unlike covalent bonds, ionic bonds are not directed at only one other atom. instead all anions, particularly those close by, are attracted to a given cation and vice versa. The oppositely charged ions are held together by strong electrostatic forces, which are non directional. therefore ionic compounds are hard, brittle due to strong forces of attraction. It is important to know how to and covert ionic formula of an ionic compound to a name and vice versa and this will help to apply solve chemical problems involving ionic compounds. Ion: an atom or molecule that gains or loses electrons (acquires an electrical charge). atoms form cations ( charge), when they lose electrons, or anions ( charge), when they gain electrons. ionic bonds are strong bonds formed when oppositely charged ions are attracted to each other.

Iconic Bonding Periodic Presidents It is important to know how to and covert ionic formula of an ionic compound to a name and vice versa and this will help to apply solve chemical problems involving ionic compounds. Ion: an atom or molecule that gains or loses electrons (acquires an electrical charge). atoms form cations ( charge), when they lose electrons, or anions ( charge), when they gain electrons. ionic bonds are strong bonds formed when oppositely charged ions are attracted to each other. Ionic bonds are characterized by the complete transfer of electrons from one atom to another, resulting in the formation of two charged particles known as ions, which are held together with the help of electrostatic forces. Pdf | short article explaining ionic bonding. | find, read and cite all the research you need on researchgate. Find the sum of valence electrons of all atoms in the polyatomic ion or molecule. if it is an anion, add one electron for each negative charge. Explain the shapes of, and bond angles in, molecules by using the qualitative model of electron pair repulsion (including lone pairs), using as simple examples: bf3 (trigonal), co2 (linear), ch4 (tetrahedral), nh3 (pyramidal), h2o (non linear), sf6 (octahedral), pf5 (trigonal bipyramid).
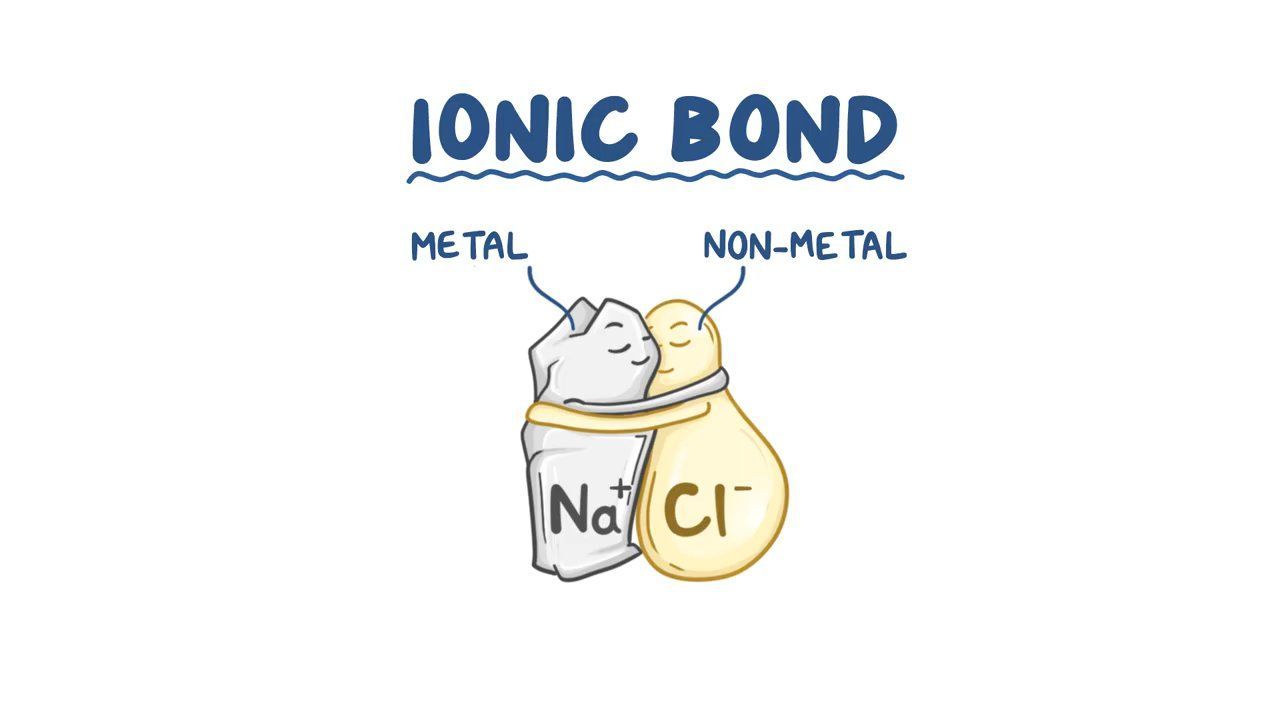
Video Ionic Bonding Osmosis Ionic bonds are characterized by the complete transfer of electrons from one atom to another, resulting in the formation of two charged particles known as ions, which are held together with the help of electrostatic forces. Pdf | short article explaining ionic bonding. | find, read and cite all the research you need on researchgate. Find the sum of valence electrons of all atoms in the polyatomic ion or molecule. if it is an anion, add one electron for each negative charge. Explain the shapes of, and bond angles in, molecules by using the qualitative model of electron pair repulsion (including lone pairs), using as simple examples: bf3 (trigonal), co2 (linear), ch4 (tetrahedral), nh3 (pyramidal), h2o (non linear), sf6 (octahedral), pf5 (trigonal bipyramid).
Comments are closed.