Ionic Bonding Definition And Examples

Ionic Bond Ionic Bonding Definition And Examples Ionic Bonding An What is an ionic bond? an ionic bond, also known as an electrovalent bond, is a type of chemical bond formed due to the electrostatic attraction between oppositely charged ions in an ionic compound. Ionic bond, type of linkage formed from the electrostatic attraction between oppositely charged ions in a chemical compound. such a bond forms when the valence (outermost) electrons of one atom are transferred permanently to another atom. learn more about ionic bonds in this article.

Ionic Bond Ionic Bonding Definition And Examples Ionic Bonding An Get the ionic bond definition and examples in chemistry. learn which types of atoms participate in ionic bonding. Ionic bonding is a form of chemical connection in which one atom loses valence electrons and another atom gains these valence electrons attaining ve and ve charges respectively. the attractive electrostatic interactions between two ions of opposite charge form an ionic bond. Learn what an ionic bond is in chemistry with clear definitions, examples, diagrams, and easy comparisons to other bond types. perfect for students!. Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, [1] and is the primary interaction occurring in ionic compounds.
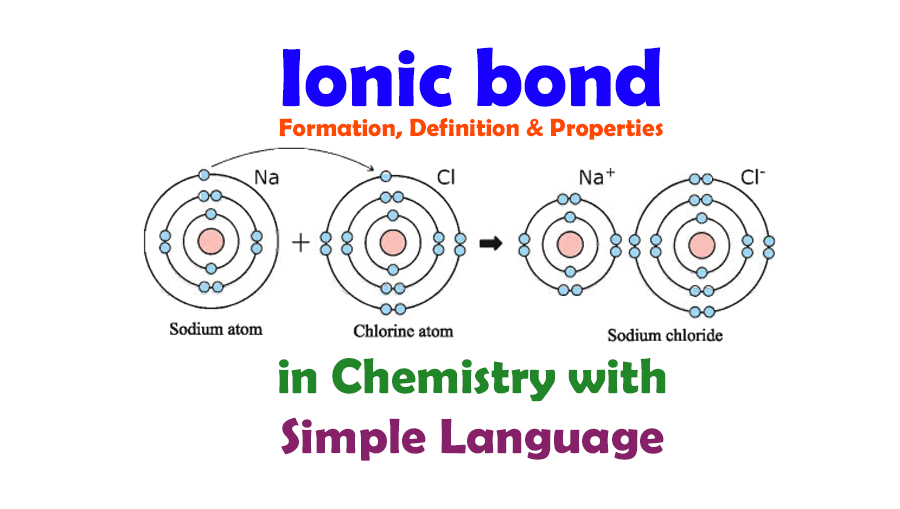
Ionic Bond Definition Properties Examples Facts 40 Off Learn what an ionic bond is in chemistry with clear definitions, examples, diagrams, and easy comparisons to other bond types. perfect for students!. Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, [1] and is the primary interaction occurring in ionic compounds. Ionic bonding is a type of chemical bond formed through the transfer of electrons from one atom to another, resulting in the formation of positively charged ions (cations) and negatively charged ions (anions). An ionic bond is one of the most important chemical bond types in chemistry. it occurs when atoms transfer electrons to achieve stable electron configurations, creating ions that attract each other through electrostatic forces. understanding ionic bonds is essential for predicting the behavior of salts, minerals, and other ionic compounds in both laboratory and everyday settings. This is one of the two main ways atoms bond to each other, the other being covalent bonding, where atoms share electrons instead. ionic bonding typically occurs between metals and nonmetals, and it produces many of the crystalline solids you encounter every day, from table salt to baking soda. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. the properties of ionic compounds shed some light on the nature of ionic bonds.

Ionic Compounds Definition Properties Examples 46 Off Ionic bonding is a type of chemical bond formed through the transfer of electrons from one atom to another, resulting in the formation of positively charged ions (cations) and negatively charged ions (anions). An ionic bond is one of the most important chemical bond types in chemistry. it occurs when atoms transfer electrons to achieve stable electron configurations, creating ions that attract each other through electrostatic forces. understanding ionic bonds is essential for predicting the behavior of salts, minerals, and other ionic compounds in both laboratory and everyday settings. This is one of the two main ways atoms bond to each other, the other being covalent bonding, where atoms share electrons instead. ionic bonding typically occurs between metals and nonmetals, and it produces many of the crystalline solids you encounter every day, from table salt to baking soda. Compounds composed of ions are called ionic compounds (or salts), and their constituent ions are held together by ionic bonds: electrostatic forces of attraction between oppositely charged cations and anions. the properties of ionic compounds shed some light on the nature of ionic bonds.
Comments are closed.