Dilute Solutions Pdf Materials Quantity

Dilute Solutions Pdf Materials Quantity Solutions are stored in a concentrated form to save space and the concentrated solutions are diluted to variable concentrations as needs result. a microscopic amount of a substance is. Dilute solutions free download as pdf file (.pdf), text file (.txt) or read online for free. this document contains 19 multiple choice questions related to solutions and colligative properties.

Thermodynamics Dilute Solutions Pdf Solution Chemical Bond It is a step wise dilution of a solution, where the dilution factor is constant at each step. the source of dilution material for each step comes from the diluted material of the previous step. The dilution factor is the total number of unit volumes in which your material will be dissolved. the diluted material must then be thoroughly mixed to achieve the true dilution. Dilution factors: ors are often used. a dilution factor is simply a multiplier that changes the final analyzed concentration back to the original concentration of th undiluted solution. dilution factors are simply the ratio of the final volume divided by ci = cf [vf vi] ,. Preparation of solutions by dilution: solutions with small concentrations are often prepared by diluting a more concentrated stock solution. a known volume of the stock solution is transferred to a new container and brought to a new volume.

Pdf Dilute Solution Colligative Properties Assignment Pdf Dokumen Dilution factors: ors are often used. a dilution factor is simply a multiplier that changes the final analyzed concentration back to the original concentration of th undiluted solution. dilution factors are simply the ratio of the final volume divided by ci = cf [vf vi] ,. Preparation of solutions by dilution: solutions with small concentrations are often prepared by diluting a more concentrated stock solution. a known volume of the stock solution is transferred to a new container and brought to a new volume. The concentration of a solution refers to the quantity of solute dissolved in a particular quantity of solvent or solution. there are several ways of expressing solution concentrations such as moles per litre, percentage concentration, grams per litre, saturated solutions etc. The amount of solute that is dissolved in a given quantity of solvent is called the concentration of the solution. a dilute solution contains only a small amount of solute in a given amount of solution, while a concentrated solution contains a large amount of solute in a given amount of solution. From the information calculated in the dilution calculation sheet, pipette the required amount of solvent into the stock bottle, along with the active and vortex. In doing dilution calculations, vd is the volume of the final, dilute solution. we add enough water (solvent) to reach this volume, just as we did in making “molar” solutions from the solid (molarity handout).
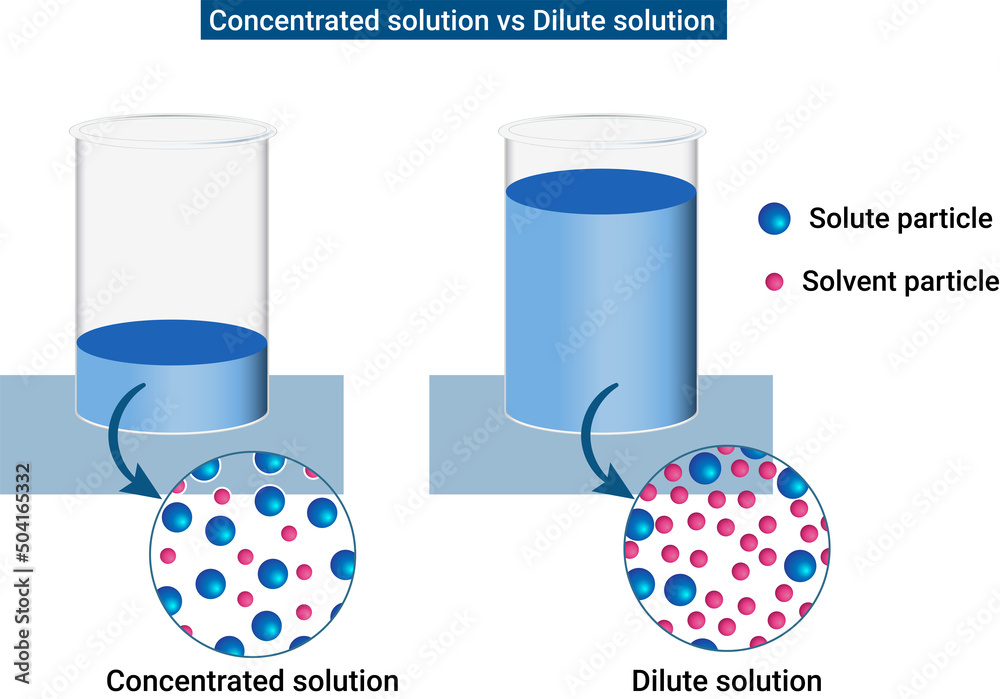
Example Of Dilute Solution In Chemistry At Victoria Brownlee Blog The concentration of a solution refers to the quantity of solute dissolved in a particular quantity of solvent or solution. there are several ways of expressing solution concentrations such as moles per litre, percentage concentration, grams per litre, saturated solutions etc. The amount of solute that is dissolved in a given quantity of solvent is called the concentration of the solution. a dilute solution contains only a small amount of solute in a given amount of solution, while a concentrated solution contains a large amount of solute in a given amount of solution. From the information calculated in the dilution calculation sheet, pipette the required amount of solvent into the stock bottle, along with the active and vortex. In doing dilution calculations, vd is the volume of the final, dilute solution. we add enough water (solvent) to reach this volume, just as we did in making “molar” solutions from the solid (molarity handout).

Dilute Solutions Pdf From the information calculated in the dilution calculation sheet, pipette the required amount of solvent into the stock bottle, along with the active and vortex. In doing dilution calculations, vd is the volume of the final, dilute solution. we add enough water (solvent) to reach this volume, just as we did in making “molar” solutions from the solid (molarity handout).
Comments are closed.