Dilute Solutions

How To Dilute Solutions 8 Steps With Pictures Wikihow A dilute solution is one in which there is a relatively small amount of solute dissolved in the solution. a concentrated solution contains a relatively large amount of solute. The act of reducing the concentration of a solution by adding additional solvent is known as dilution. dilution involves adding additional solvent to a sample of a solution. the process does not involve a change in the amount of solute. it is the volume of the solution that changes.

How To Dilute Solutions 8 Steps With Pictures Wikihow A dilute solution contains a relatively small amount of solute dissolved in a comparatively large volume of solvent. this means the solute particles are spread out significantly within the solvent. This video explains the process of dilution through real world examples and demonstrates how to use the m1v1=m2v2 formula to prepare laboratory solutions fro. At its heart, dilution is simple: adding a solvent (the diluent, usually water) to a concentrated solution (the stock) to reduce its strength. the classic formula everyone learns is c₁v₁ = c₂v₂, where c is concentration and v is volume. Dilution is the process of “lowering the concentration of a solute in a solution by simply adding more solvent to the solution, such as water.” diluting a solution entails adding more solvent without adding more solute.

How To Dilute Solutions 8 Steps With Pictures Wikihow At its heart, dilution is simple: adding a solvent (the diluent, usually water) to a concentrated solution (the stock) to reduce its strength. the classic formula everyone learns is c₁v₁ = c₂v₂, where c is concentration and v is volume. Dilution is the process of “lowering the concentration of a solute in a solution by simply adding more solvent to the solution, such as water.” diluting a solution entails adding more solvent without adding more solute. Dilution involves adding more solvent, typically water, to a solution to decrease its concentration. for instance, when a dark purple solution is gradually mixed with water, the color lightens to a fuchsia hue, indicating a reduction in concentration. Dilution you are a work study student in our chemistry department. amy, your supervisor has just asked you to prepare 100ml of 2.00m h 2 so 4 solution for tomorrow's undergraduate experiment. the stock solutions cabinet is under the stockroom explorer. you will find find a 2.50 liter bottle containing 17.8m h 2 so 4. Free dilution calculator using c1v1=c2v2. solve for initial concentration, initial volume, final concentration, or final volume. find how much solvent to add for any dilution. Diluting a solvent means to lower the concentration by adding more of the solvent to the solution. this can be done by adding solvent to the main bulk of the solution or taking some volume of the solution and adding solvent to it.
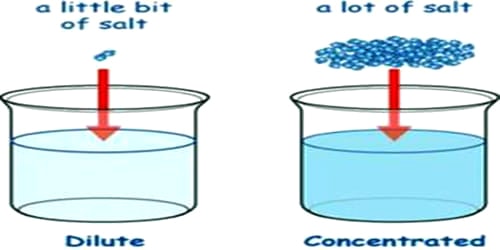
Experiment Differentiate Between Concentrated And Dilute Solutions Dilution involves adding more solvent, typically water, to a solution to decrease its concentration. for instance, when a dark purple solution is gradually mixed with water, the color lightens to a fuchsia hue, indicating a reduction in concentration. Dilution you are a work study student in our chemistry department. amy, your supervisor has just asked you to prepare 100ml of 2.00m h 2 so 4 solution for tomorrow's undergraduate experiment. the stock solutions cabinet is under the stockroom explorer. you will find find a 2.50 liter bottle containing 17.8m h 2 so 4. Free dilution calculator using c1v1=c2v2. solve for initial concentration, initial volume, final concentration, or final volume. find how much solvent to add for any dilution. Diluting a solvent means to lower the concentration by adding more of the solvent to the solution. this can be done by adding solvent to the main bulk of the solution or taking some volume of the solution and adding solvent to it.
Comments are closed.