Dilute Solution
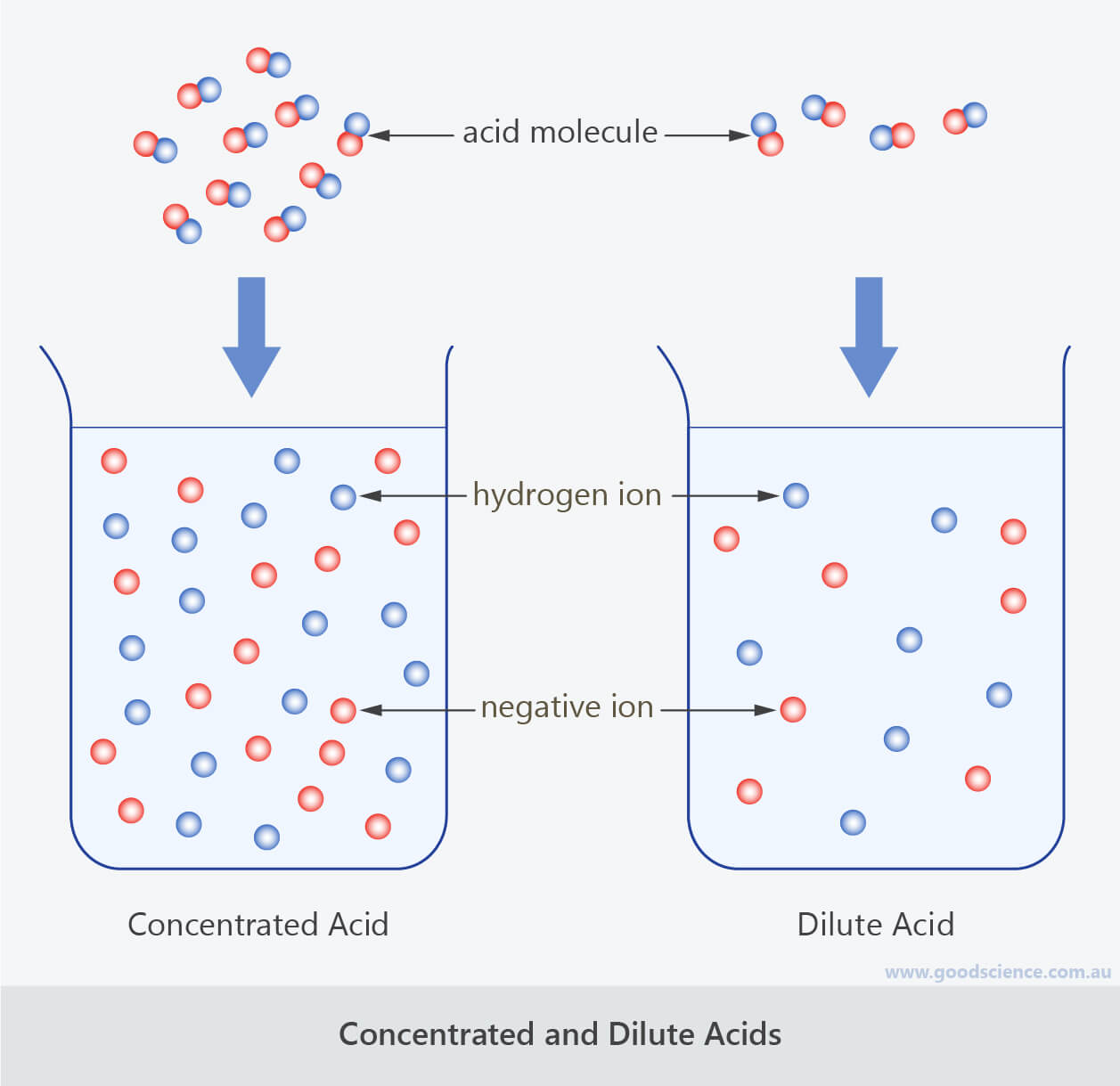
Dilute Solution Learn how to change concentrations of solutions by dilution or concentration. find out how to use stock solutions, volumetric flasks, and dilution equations. A dilute solution contains a relatively small amount of solute dissolved in a comparatively large volume of solvent. this means the solute particles are spread out significantly within the solvent.

Dilute Solution The act of reducing the concentration of a solution by adding additional solvent is known as dilution. dilution involves adding additional solvent to a sample of a solution. the process does not involve a change in the amount of solute. it is the volume of the solution that changes. Learn how to accurately dilute solutions with this simple guide. we explain how to calculate dilutions with a formula and how to make quick dilutions. A dilute solution is a fundamental concept in chemistry and engineering, describing a mixture where the dissolved substance is spread out thinly within the liquid. A dilute solution is a type of solution where the concentration of the solute is relatively low compared to the solvent. this means the solution contains a small amount of dissolved substance dispersed throughout a larger volume of the solvent.

Dilute Solution Pdf A dilute solution is a fundamental concept in chemistry and engineering, describing a mixture where the dissolved substance is spread out thinly within the liquid. A dilute solution is a type of solution where the concentration of the solute is relatively low compared to the solvent. this means the solution contains a small amount of dissolved substance dispersed throughout a larger volume of the solvent. Learn how to calculate the molarity of a dilute solution by adding more solvent without more solute. see examples of dilution problems with solutions, equations and answers. A dilute solution is one that has a relatively small amount of dissolved solute. however, these terms are relative, and we need to be able to express concentration in a more exact, quantitative manner. A dilute solution is one in which the amount of solute (the substance that is dissolved) is relatively low compared to the amount of solvent (the substance in which the solute is dissolved). in other words, a dilute solution contains a small amount of solute relative to the solvent. Learn what dilution is, how to calculate it using a formula, and how to perform simple and serial dilutions. find out why dilution is important in chemistry, biology and other fields.

Dilute Solution Pdf Learn how to calculate the molarity of a dilute solution by adding more solvent without more solute. see examples of dilution problems with solutions, equations and answers. A dilute solution is one that has a relatively small amount of dissolved solute. however, these terms are relative, and we need to be able to express concentration in a more exact, quantitative manner. A dilute solution is one in which the amount of solute (the substance that is dissolved) is relatively low compared to the amount of solvent (the substance in which the solute is dissolved). in other words, a dilute solution contains a small amount of solute relative to the solvent. Learn what dilution is, how to calculate it using a formula, and how to perform simple and serial dilutions. find out why dilution is important in chemistry, biology and other fields.
Comments are closed.