Crystallisation Labelled Diagram

Crystallisation Diagram Drag and drop the pins to their correct place on the image water , salt solution, heat, tripod, gauze, evaporating basin, beaker. Crystallization, a fundamental process in nature and industry, is pivotal in forming structured solids from liquids or gases. it involves the orderly arrangement of atoms, ions, or molecules into a highly organized, repeating pattern known as a crystal lattice.
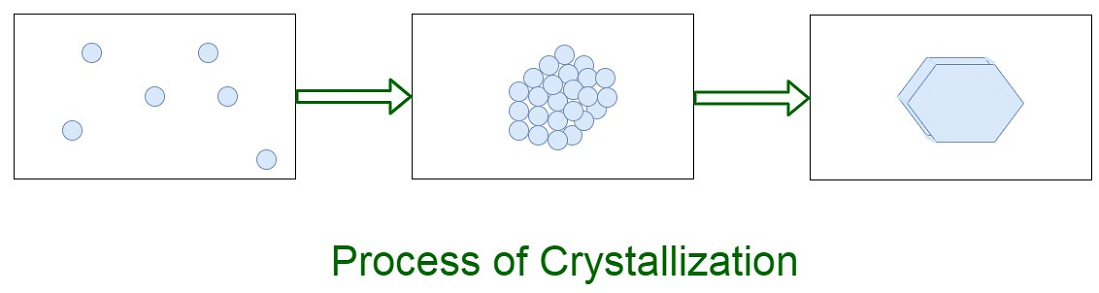
Crystallisation Diagram Need a crystallisation diagram? this illustration of the process of crystallisation can be printed for students to label and add their own information to. This illustration of the process of crystallisation can be printed, cut and stuck into exercise books for students to label and add their own information to. Figure 3.1: various crystals. crystallization is used in the chemistry laboratory as a purification technique for solids. an impure solid is completely dissolved in a minimal amount of hot, boiling solvent, and the hot solution is allowed to slowly cool. These diagrams map the specific conditions under which crystallization occurs. they allow for the prediction of when and how a material will solidify, which is valuable in many scientific and industrial processes.
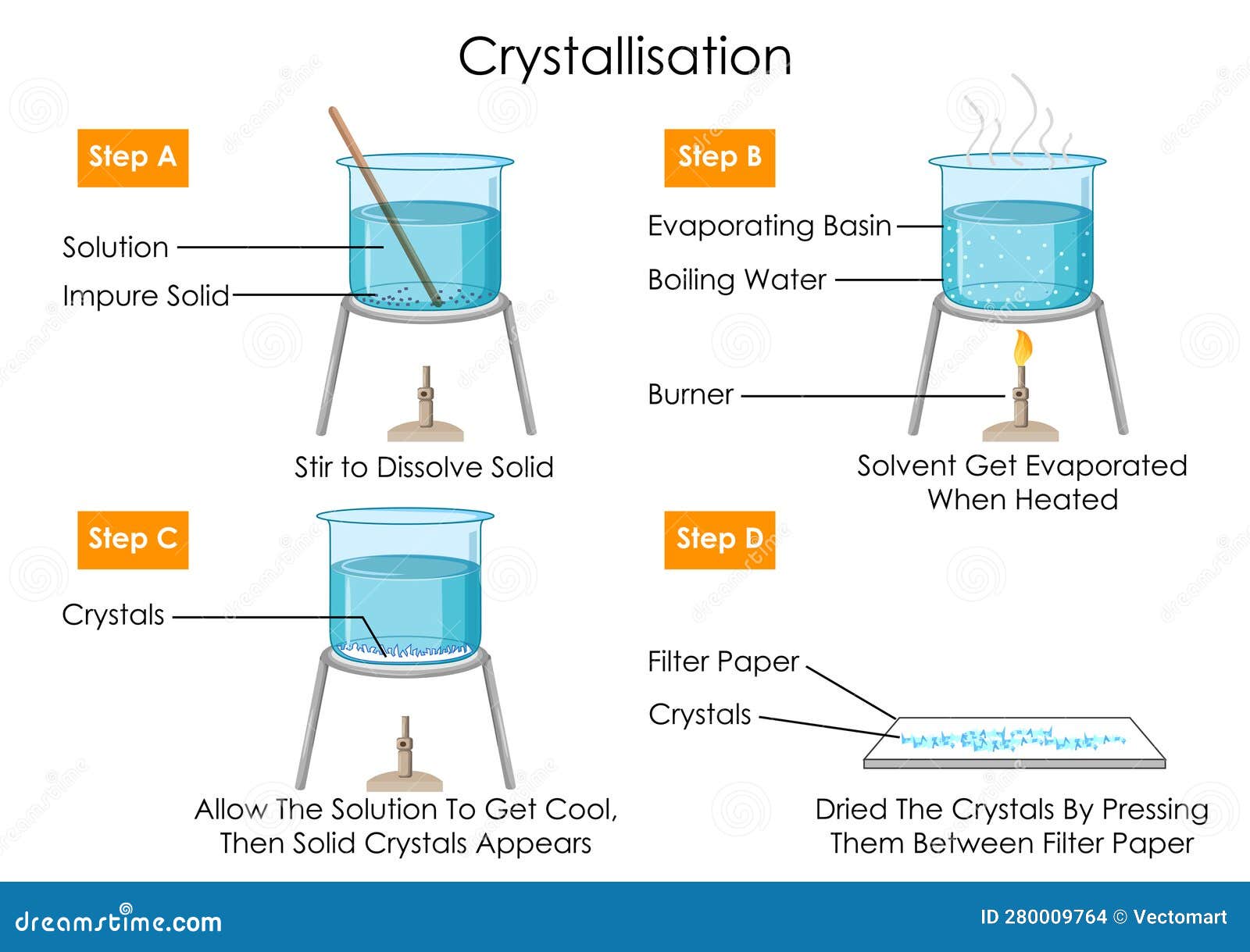
Crystallisation Diagram Vd Noxtals Jpg Figure 3.1: various crystals. crystallization is used in the chemistry laboratory as a purification technique for solids. an impure solid is completely dissolved in a minimal amount of hot, boiling solvent, and the hot solution is allowed to slowly cool. These diagrams map the specific conditions under which crystallization occurs. they allow for the prediction of when and how a material will solidify, which is valuable in many scientific and industrial processes. Schematic showing various crystallisation mechanisms on surfaces. circles represent ions, rectangles represent crystals and irregular shapes represent an amorphous phase. The solidification of a liquid material into a strongly ordered solid whose atoms or molecules are arranged in a well defined three dimensional crystal lattice is known as crystallisation. This first series of diagrams shows what happens if you let a crystallization proceed slowly: first by setting the flask at room temperature undisturbed until crystals form, and then carefully on ice. the red bar to the right of each image is a thermometer, to indicate the temperature. Crystallisation is a technique that is used to separate a soluble solute from a solution. crystallisation involves the formation of a saturated solution and temperature differences. crystallisation results in the loss of the solvent to the surroundings.

Crystallisation Diagram Vd Noxtals Jpg Schematic showing various crystallisation mechanisms on surfaces. circles represent ions, rectangles represent crystals and irregular shapes represent an amorphous phase. The solidification of a liquid material into a strongly ordered solid whose atoms or molecules are arranged in a well defined three dimensional crystal lattice is known as crystallisation. This first series of diagrams shows what happens if you let a crystallization proceed slowly: first by setting the flask at room temperature undisturbed until crystals form, and then carefully on ice. the red bar to the right of each image is a thermometer, to indicate the temperature. Crystallisation is a technique that is used to separate a soluble solute from a solution. crystallisation involves the formation of a saturated solution and temperature differences. crystallisation results in the loss of the solvent to the surroundings.
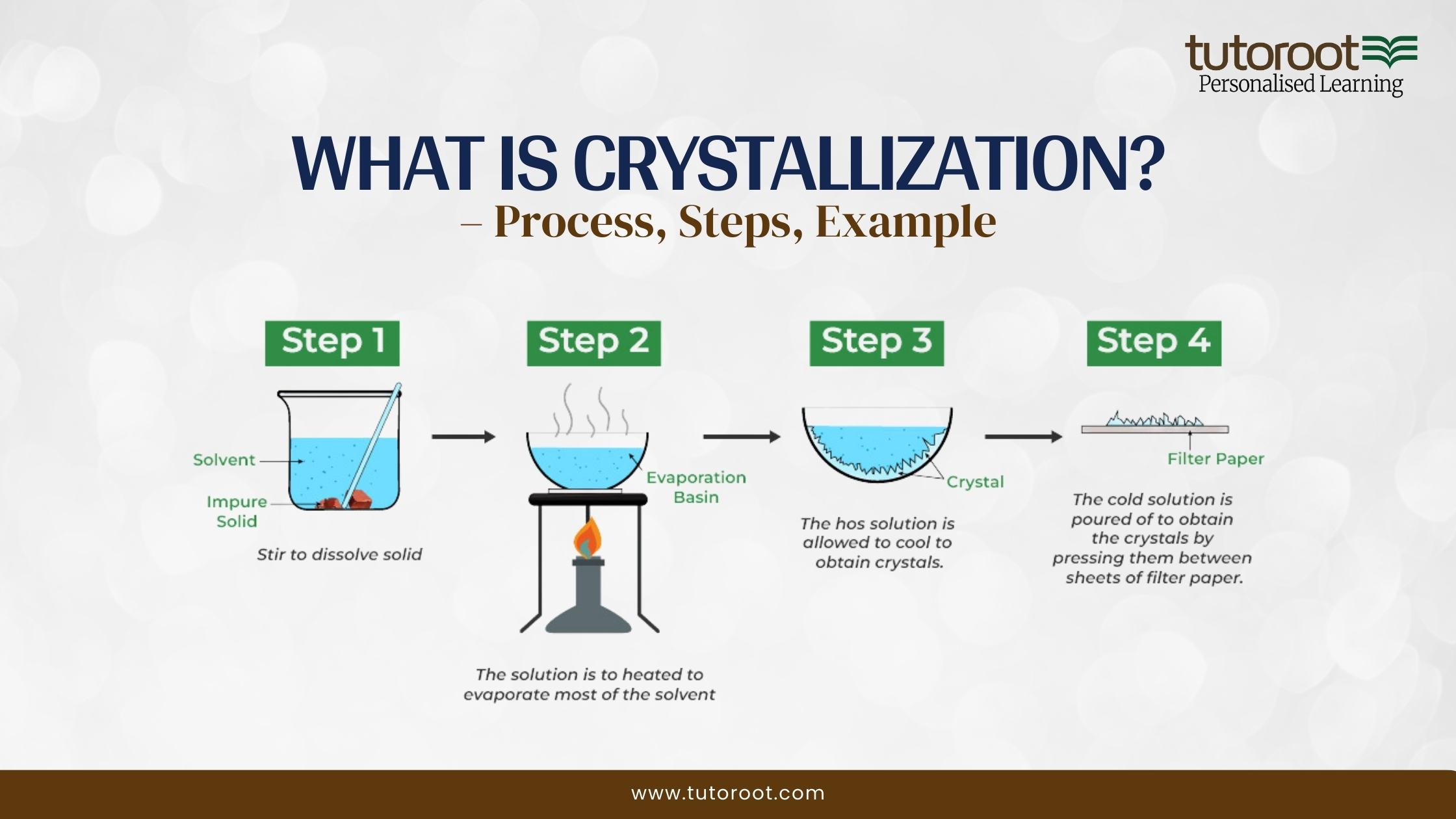
Crystallisation Diagram Vd Noxtals Jpg This first series of diagrams shows what happens if you let a crystallization proceed slowly: first by setting the flask at room temperature undisturbed until crystals form, and then carefully on ice. the red bar to the right of each image is a thermometer, to indicate the temperature. Crystallisation is a technique that is used to separate a soluble solute from a solution. crystallisation involves the formation of a saturated solution and temperature differences. crystallisation results in the loss of the solvent to the surroundings.
Comments are closed.