What Is Crystallisation

Document Moved Crystallization is the formation of ordered, repeating patterns of atoms, ions, or molecules from liquids or gases. learn how nucleation, growth, and aggregation shape crystallization, and see examples from nature and industry. Crystallization is the process of forming a solid substance with a well defined three dimensional lattice from a solution. learn how crystallization works, what factors affect it, and what are its uses and examples with byju's.

Crystallisation Vamtec Crystallization is a process that leads to solids with a uniform pattern of atoms or molecules, i.e. a crystal. the uniform nature of a crystalline solid can be contrasted with amorphous solids in which atoms or molecules lack regular organization. Solids that appear insoluble in a solvent do in fact have a (normally small) portion of material that dissolves. crystallization is most common with solids that have moderate solubility at low temperatures, so that heat can "tip them over the edge" to completely dissolve. Crystallization is the process of organizing atoms or molecules into a well defined, rigid crystal lattice to minimize their energy state. learn about the four types of crystallization, the steps involved, and the applications in various industries. Crystallization is a technique for purification of the substances. it is a technique that separates a solid from its solution. the process in which the solid dissolve in the liquid arranges itself in well defined 3 d structures called the crystal is called crystallization.

Crystallisation Crystallization is the process of organizing atoms or molecules into a well defined, rigid crystal lattice to minimize their energy state. learn about the four types of crystallization, the steps involved, and the applications in various industries. Crystallization is a technique for purification of the substances. it is a technique that separates a solid from its solution. the process in which the solid dissolve in the liquid arranges itself in well defined 3 d structures called the crystal is called crystallization. Crystallization involves the transformation of atoms or molecules from a fluid or gaseous state into a highly organized solid structure known as a crystal. unlike amorphous solids, which lack a defined internal arrangement, crystals possess a precise, repeating three dimensional pattern. Crystallization is a widely used separation technique in chemistry, essential for obtaining pure solid substances from a solution, melt, or vapor phase. Crystallization, or crystallisation, is the process of atoms or molecules arranging into a well defined, rigid crystal lattice in order to minimize their energetic state. The freezing process, also called crystallization, involves cooling salt water to form crystals of pure ice. the ice crystals are separated from the unfrozen brine, rinsed to remove residual salt, and then melted to produce fresh water. freezing is theoretically more efficient than distillation, and scaling as well as corrosion….
/GettyImages-1130233838-2ec5c241a6344098a97f9311bba023d4.jpg)
Crystallisation Definition Crystallization involves the transformation of atoms or molecules from a fluid or gaseous state into a highly organized solid structure known as a crystal. unlike amorphous solids, which lack a defined internal arrangement, crystals possess a precise, repeating three dimensional pattern. Crystallization is a widely used separation technique in chemistry, essential for obtaining pure solid substances from a solution, melt, or vapor phase. Crystallization, or crystallisation, is the process of atoms or molecules arranging into a well defined, rigid crystal lattice in order to minimize their energetic state. The freezing process, also called crystallization, involves cooling salt water to form crystals of pure ice. the ice crystals are separated from the unfrozen brine, rinsed to remove residual salt, and then melted to produce fresh water. freezing is theoretically more efficient than distillation, and scaling as well as corrosion….
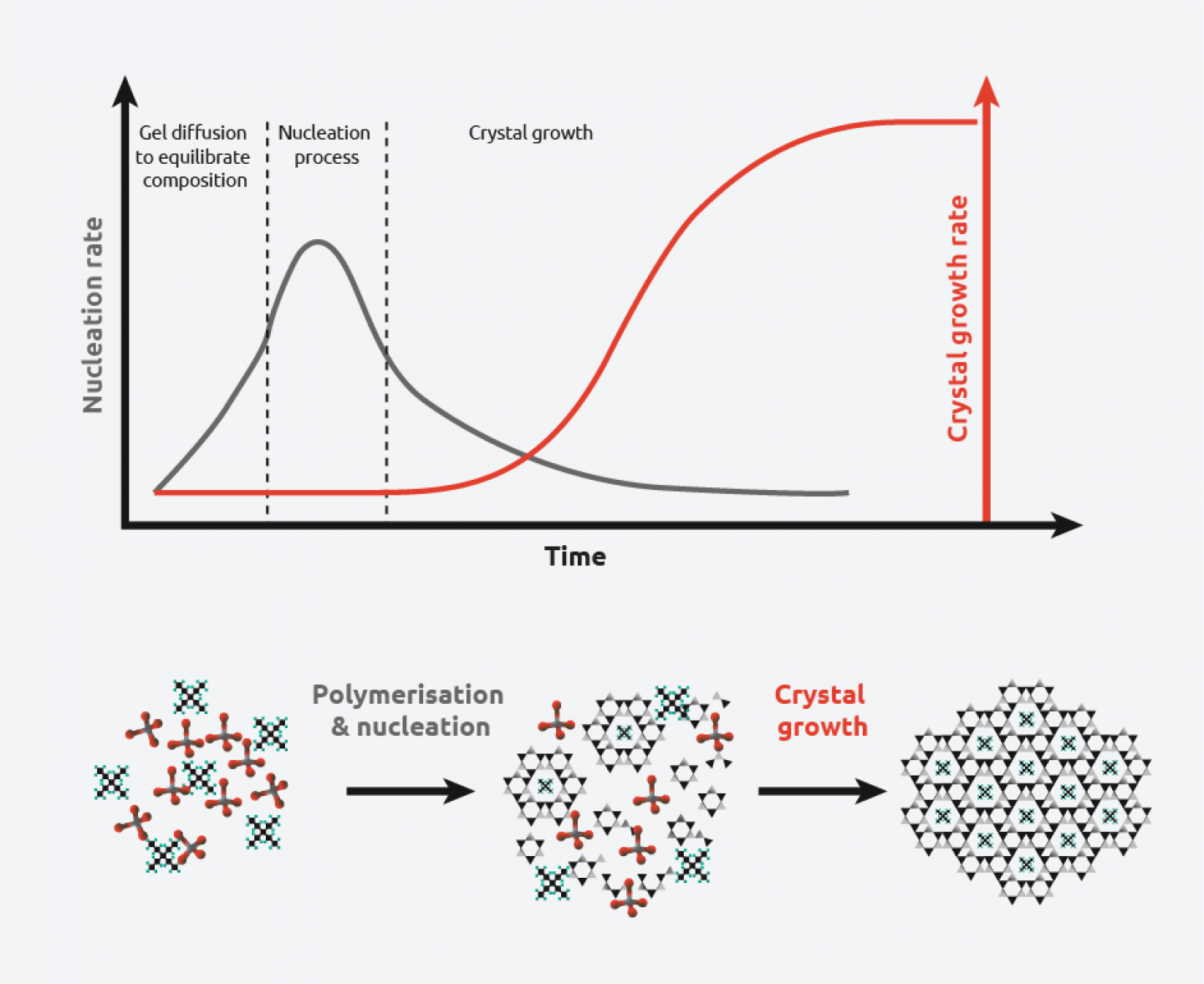
Crystallisation Definition Crystallization, or crystallisation, is the process of atoms or molecules arranging into a well defined, rigid crystal lattice in order to minimize their energetic state. The freezing process, also called crystallization, involves cooling salt water to form crystals of pure ice. the ice crystals are separated from the unfrozen brine, rinsed to remove residual salt, and then melted to produce fresh water. freezing is theoretically more efficient than distillation, and scaling as well as corrosion….

Crystallisation Crystallization Organic Chemistry Chemistry
Comments are closed.