Chem102 Ch15 Pt5 Example

5 Part 2 Chm 101 Pdf Enjoy the videos and music you love, upload original content, and share it all with friends, family, and the world on . Additional example #1 first set up the ice table and add the values in the problem ch4(g) ⇌ c2h2(g) 3 h2(g).

Chem 102 Tutorial 5 Solutions Pdf Mole Unit Scientific Phenomena It defines equilibrium as a state where competing processes are balanced so that no observable change occurs over time. it discusses different types of equilibrium, including physical, chemical, and dynamic equilibrium. Example 15 judging whether a mixture is at equilibrium the following pictures represent mixtures of a molecules (red spheres) and b molecules (blue spheres), which interconvert according to the equation a b. Quizlet has study tools to help you learn anything. improve your grades and reach your goals with flashcards, practice tests and expert written solutions today. Subjects chem 102 chapter 15 exam questions with complete s reversible process answer occurs when the produ.
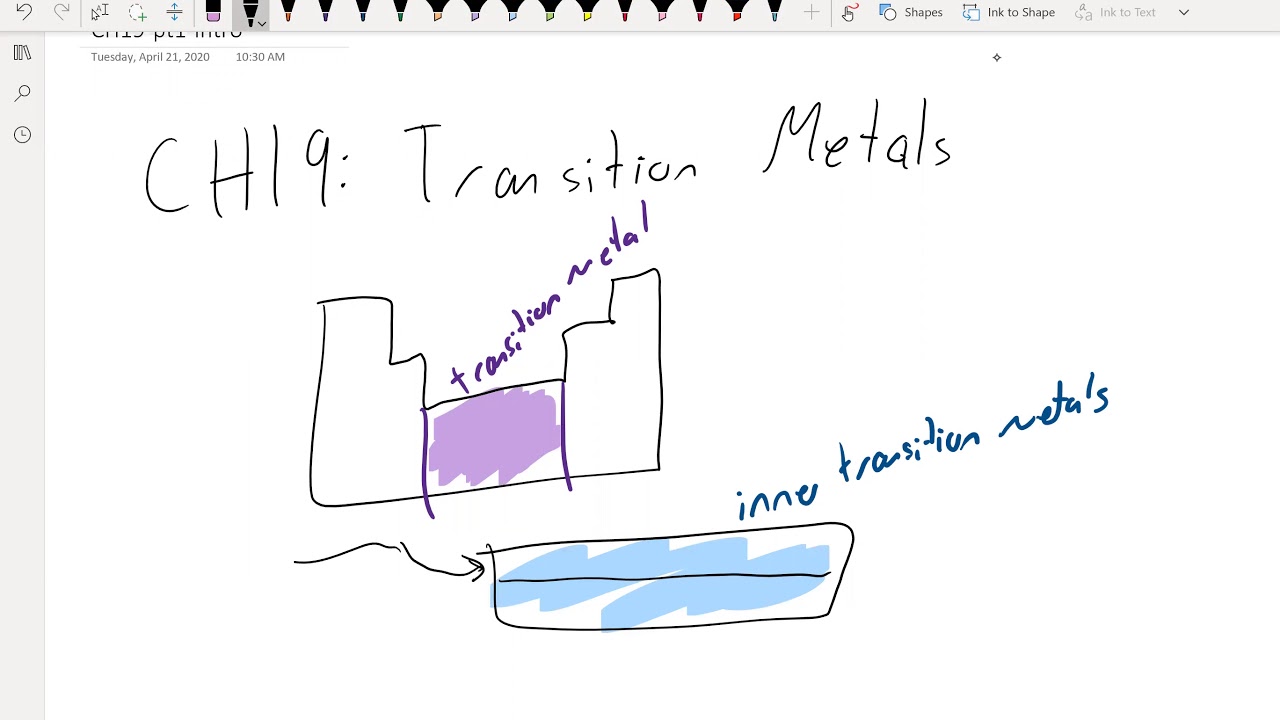
Chem102 Ch19 Pt1 Intro Youtube Quizlet has study tools to help you learn anything. improve your grades and reach your goals with flashcards, practice tests and expert written solutions today. Subjects chem 102 chapter 15 exam questions with complete s reversible process answer occurs when the produ. Share your videos with friends, family, and the world. Chem 102 lab practice exam answer key free download as pdf file (.pdf) or read online for free. Name megan rizzo lab partner emma kurtz unc onyen mrizzo22 experiment 15.2: post lab assignment (80 total points) instructions: 1. parts of this post lab assignment will be completed during lab (data entry and observations) and parts are to be completed at home (questions, calculations, and figures). 2. These are a mixture of problems from the book and others collected. instructions: indicate answers in the provided blanks. on a separate sheet of paper, show work. if work is not. 1. write the expression for kc for the following reactions. a. n 2 (g) o 2 (g) ⇌ no (g) b. ti (s) cl 2 (g) ⇌ ticl 4 (l) d. feo (s) h 2 (g) ⇌ fe (s) h 2 o (l) e.
Comments are closed.