Chem102 Ch15 Pt10 Example Youtube
Ch 10 Youtube Enjoy the videos and music you love, upload original content, and share it all with friends, family, and the world on . Share your videos with friends, family, and the world.

Ch10 Video1 Youtube Enjoy the videos and music you love, upload original content, and share it all with friends, family, and the world on . Share your videos with friends, family, and the world. Coordination compounds crystal field theory, part 2, including pex 4 & 5. Chem 102: general chemistry i (chang et al. 13th edition) dr. emily caudill · course.

Chem P 10 Youtube Coordination compounds crystal field theory, part 2, including pex 4 & 5. Chem 102: general chemistry i (chang et al. 13th edition) dr. emily caudill · course. How to calculate molecular weight (mwt) of compound ?. Share your videos with friends, family, and the world. Prepare for the chem 102 midterm exam 1 with guidelines and sample questions covering kinetics and equilibrium. ace your chemistry test!. These are a mixture of problems from the book and others collected. instructions: indicate answers in the provided blanks. on a separate sheet of paper, show work. if work is not. 1. write the expression for kc for the following reactions. a. n 2 (g) o 2 (g) ⇌ no (g) b. ti (s) cl 2 (g) ⇌ ticl 4 (l) d. feo (s) h 2 (g) ⇌ fe (s) h 2 o (l) e.
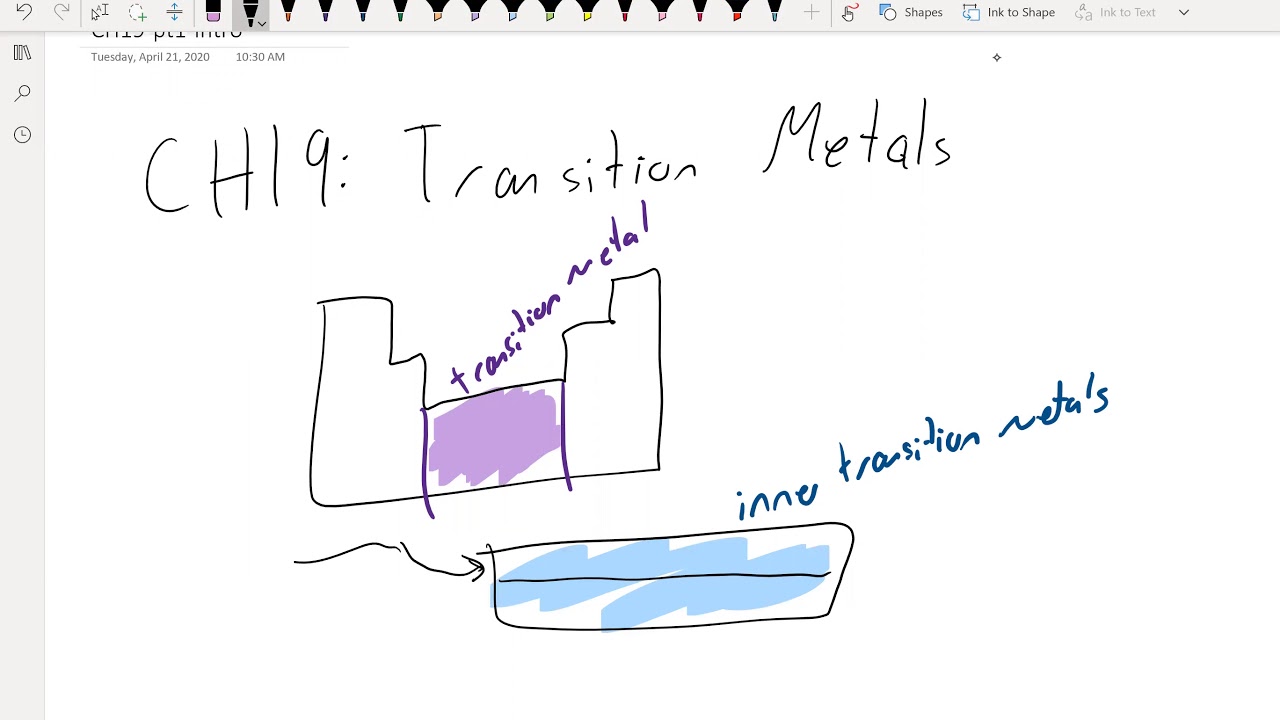
Chem102 Ch19 Pt1 Intro Youtube How to calculate molecular weight (mwt) of compound ?. Share your videos with friends, family, and the world. Prepare for the chem 102 midterm exam 1 with guidelines and sample questions covering kinetics and equilibrium. ace your chemistry test!. These are a mixture of problems from the book and others collected. instructions: indicate answers in the provided blanks. on a separate sheet of paper, show work. if work is not. 1. write the expression for kc for the following reactions. a. n 2 (g) o 2 (g) ⇌ no (g) b. ti (s) cl 2 (g) ⇌ ticl 4 (l) d. feo (s) h 2 (g) ⇌ fe (s) h 2 o (l) e.

Chem102 Ch 17 Part 4 Youtube Prepare for the chem 102 midterm exam 1 with guidelines and sample questions covering kinetics and equilibrium. ace your chemistry test!. These are a mixture of problems from the book and others collected. instructions: indicate answers in the provided blanks. on a separate sheet of paper, show work. if work is not. 1. write the expression for kc for the following reactions. a. n 2 (g) o 2 (g) ⇌ no (g) b. ti (s) cl 2 (g) ⇌ ticl 4 (l) d. feo (s) h 2 (g) ⇌ fe (s) h 2 o (l) e.
Comments are closed.