2022 2023 Random Validation Phase 2 Process Overview
The Lifecycle Approach To Process Validation Overview Tv Tpi This voiced over powerpoint video will provide leas with an overview of the 2022 2023 essa program monitoring – phase 2 random validation process conducted b. 2022 2023 program monitoring – random validations process overview 2022. texas education agency. all rights reserved.

Implementing A 2 Stage Process Validation Program Deadline to submit documentation via the fpc random validation workapp system. tea program staff complete preliminary reviews. tea contacts esc regarding leas that receive “improvement needed” status for esc review and comment before tea notifies leas. fpc rv workapp lea contacts receive notification of validation results. Federal program compliance division 2022 2023 phase 2 random validation timeline phase 2 programs x title i, part d – prevention and intervention for delinquent, neglected, and at risk youth x title v, part b – rural and low title. All resources for phase 2 of the 2022 2023 essa program monitoring – random validations process have been posted on the essa random validation monitoring webpage. This voiced over powerpoint video will provide leas with an overview of the 2021 2022 essa program monitoring – phase 2 random validation process conducted b.

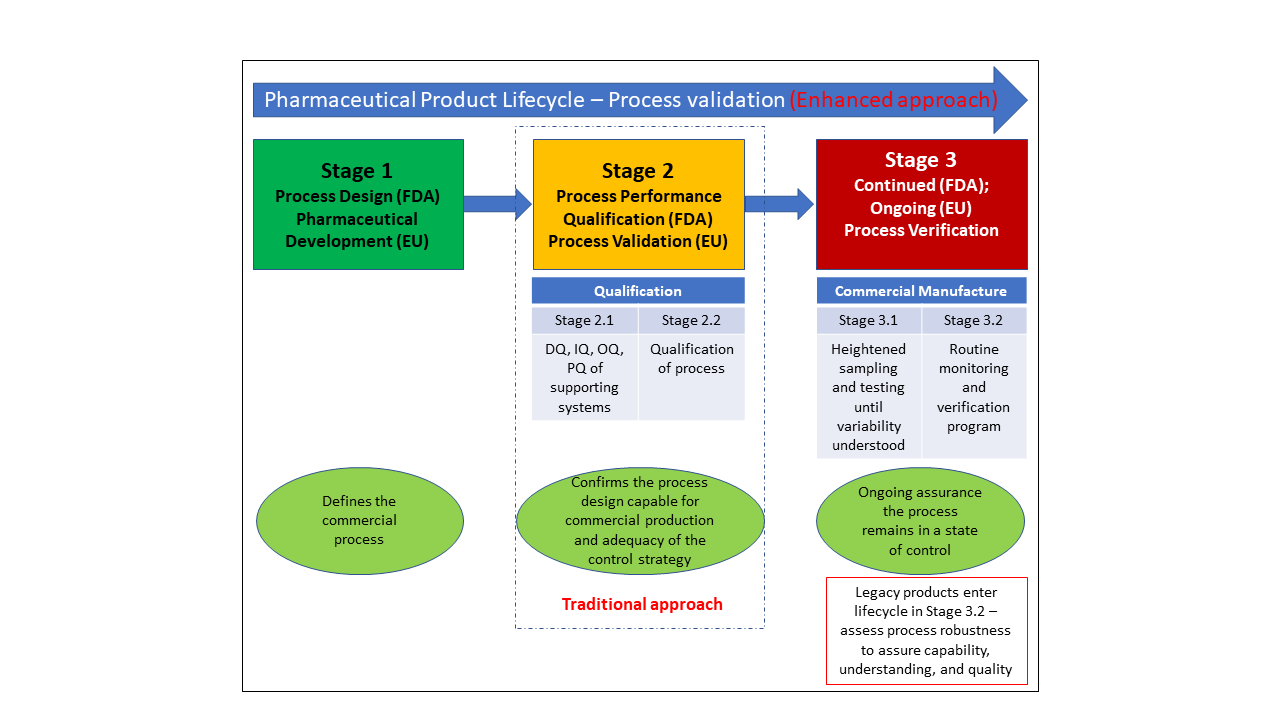
Overview Of The Three Phase Validation Study Download Scientific Diagram All resources for phase 2 of the 2022 2023 essa program monitoring – random validations process have been posted on the essa random validation monitoring webpage. This voiced over powerpoint video will provide leas with an overview of the 2021 2022 essa program monitoring – phase 2 random validation process conducted b. The federal program compliance (fpc) division will launch the 2022 2023 program monitoring random validations process in early october 2022. the random validations process framework (i.e., 2 phase process, programs, etc.) that was utilized in 2021 2022 will remain the same for 2022 2023. In the second phase, model validation, a validation set with independent samples is used for validation of the model. for identification libraries, validation involves analysing samples (i.e., challenge samples) not represented in the library to demonstrate the discriminative ability of the library model. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich assembly to the regulatory authorities of the ich regions for internal and external consultation, according to national or regional procedures. Expert consensus is particularly united on the utility of this randomized phase ii to phase iii pathway for trials with time to event endpoints, disease processes with unclear natural histories, and those with biomarker guided designs.

Overview Of The Three Phase Validation Study Download Scientific Diagram The federal program compliance (fpc) division will launch the 2022 2023 program monitoring random validations process in early october 2022. the random validations process framework (i.e., 2 phase process, programs, etc.) that was utilized in 2021 2022 will remain the same for 2022 2023. In the second phase, model validation, a validation set with independent samples is used for validation of the model. for identification libraries, validation involves analysing samples (i.e., challenge samples) not represented in the library to demonstrate the discriminative ability of the library model. At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich assembly to the regulatory authorities of the ich regions for internal and external consultation, according to national or regional procedures. Expert consensus is particularly united on the utility of this randomized phase ii to phase iii pathway for trials with time to event endpoints, disease processes with unclear natural histories, and those with biomarker guided designs.
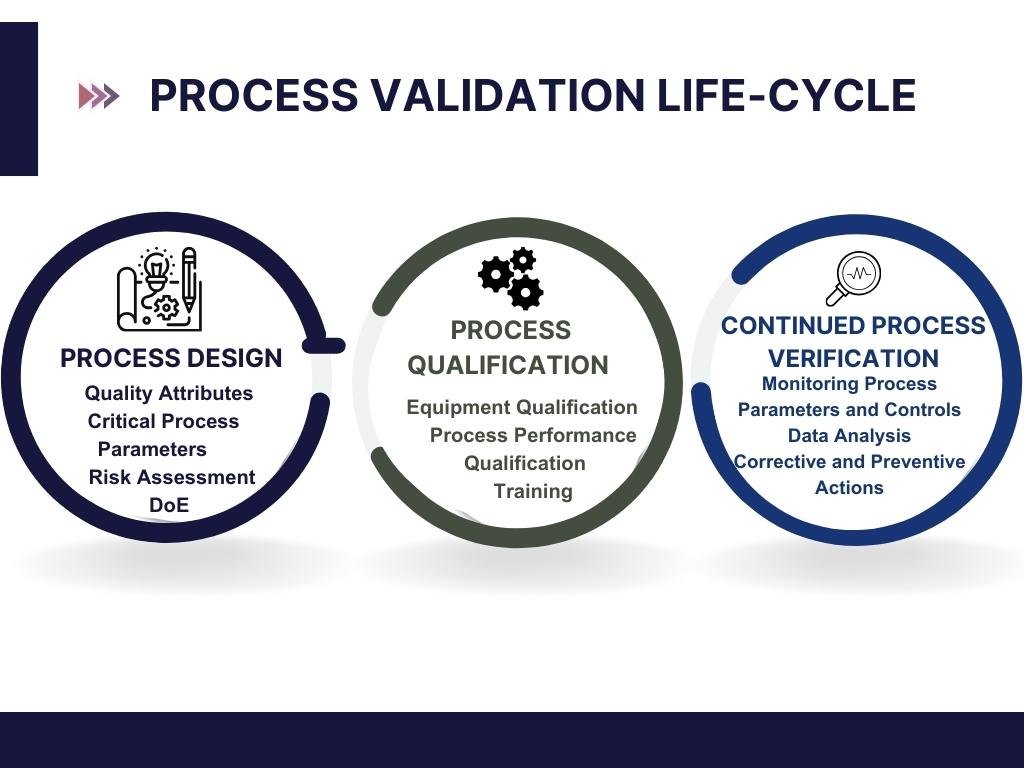
Validation Phase Drive Efficiency And Reduce Risk In Validation With A At step 2 of the ich process, a consensus draft text or guideline, agreed by the appropriate ich expert working group, is transmitted by the ich assembly to the regulatory authorities of the ich regions for internal and external consultation, according to national or regional procedures. Expert consensus is particularly united on the utility of this randomized phase ii to phase iii pathway for trials with time to event endpoints, disease processes with unclear natural histories, and those with biomarker guided designs.
Comments are closed.