Zepbound Package Insert Prescribing Information Moa

Zepbound Approved For Chronic Weight Management Clinical Advisor Zepbound is a glucose dependent insulinotropic polypeptide (gip) receptor and glucagon like peptide 1 (glp 1) receptor agonist indicated in combination with a reduced calorie diet and increased physical activity:. In rats, tirzepatide causes thyroid c cell tumors. it is unknown whether zepbound causes thyroid c cell tumors, including medullary thyroid carcinoma (mtc), in humans as the human relevance of tirzepatide induced rodent thyroid c cell tumors has not been determined (5.1, 13.1).

108074395 1733921894135 01 Ro 1920x1080 Png V 1733921969 W 1920 H 1080 Advise patients using oral hormonal contraceptives to switch to a non oral contraceptive method, or add a barrier method of contraception for 4 weeks after initiation with zepbound and for 4. Counsel patients regarding the potential risk for mtc with the use of zepbound and inform them of symptoms of thyroid tumors (e.g., a mass in the neck, dysphagia, dyspnea, persistent hoarseness). Highlights of prescribing information these highlights do not include all the information needed to use zepbound safely and effectively. see full prescribing information for zepbound. Advise patients using oral hormonal contraceptives to switch to a non oral contraceptive method, or add a barrier method of contraception for 4 weeks after initiation with zepbound and for 4.
:max_bytes(150000):strip_icc()/Eli_Lilly_and_Company_Zepbound_Vials-70e9624be6dd4cf9832e532cc778d27d.jpg)
What To Know About The New Lower Cost Version Of Zepbound Highlights of prescribing information these highlights do not include all the information needed to use zepbound safely and effectively. see full prescribing information for zepbound. Advise patients using oral hormonal contraceptives to switch to a non oral contraceptive method, or add a barrier method of contraception for 4 weeks after initiation with zepbound and for 4. Food and drug administration. Administer zepbound once weekly at any time of day, with or without meals. zepbound should not be administered daily. inject zepbound subcutaneously in the abdomen, thigh, or upper arm. rotate injection sites with each dose. zepbound should not be administered intramuscularly or intravenously. Highlights of prescribing information these highlights do not include all the information needed to use zepbound safely and effectively. see full prescribing information for zepbound. In a pool of two zepbound clinical trials for weight reduction (studies 1 and 2), severe gastrointestinal adverse reactions were reported more frequently among patients receiving zepbound (5 mg.

Fda Label For Zepbound Injection Solution Subcutaneous Indications Food and drug administration. Administer zepbound once weekly at any time of day, with or without meals. zepbound should not be administered daily. inject zepbound subcutaneously in the abdomen, thigh, or upper arm. rotate injection sites with each dose. zepbound should not be administered intramuscularly or intravenously. Highlights of prescribing information these highlights do not include all the information needed to use zepbound safely and effectively. see full prescribing information for zepbound. In a pool of two zepbound clinical trials for weight reduction (studies 1 and 2), severe gastrointestinal adverse reactions were reported more frequently among patients receiving zepbound (5 mg.
Zepbound Package Insert Drugs Highlights of prescribing information these highlights do not include all the information needed to use zepbound safely and effectively. see full prescribing information for zepbound. In a pool of two zepbound clinical trials for weight reduction (studies 1 and 2), severe gastrointestinal adverse reactions were reported more frequently among patients receiving zepbound (5 mg.
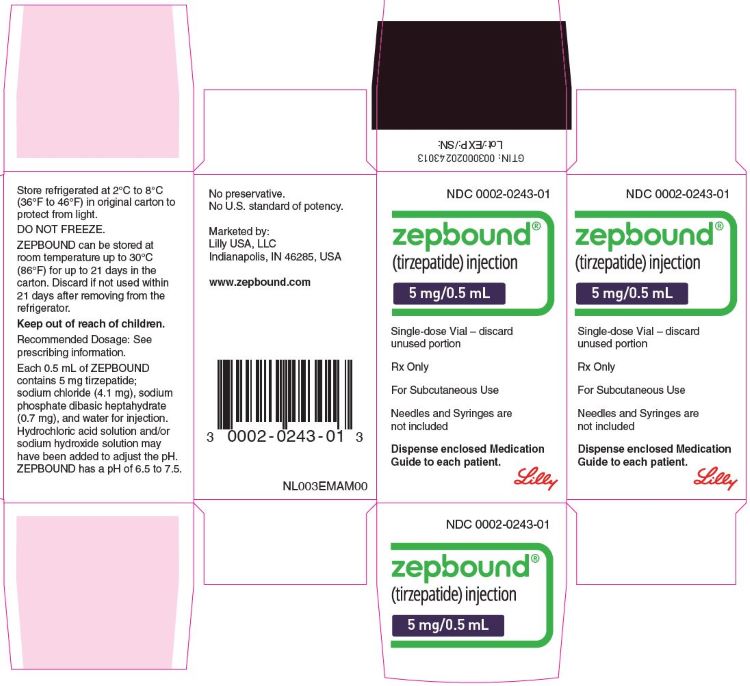
Zepbound Package Insert Drugs
Comments are closed.