Zeolites Explained And Simplified

Zeolites Chemistry Encyclopedia Structure Reaction Water Uses The word "zeolite" comes from the greek for "boiling stone," because of the early observation that zeolites release water when heated. as their compositions are not fixed, they are examples of nonstoichiometric compounds. An easy to understand introduction to zeolites, how they form, how they work as ion exchangers, molecular sieves, and catalysts, and how we use them.
The Basics Of The Structural Chemistry Of Zeolites Explained Zeolites Zeolite, any member of a family of hydrated aluminosilicate minerals that contain alkali and alkaline earth metals. the zeolites are noted for their lability toward ion exchange and reversible dehydration. learn more about the characteristics and structure of zeolites in this article. This article provides a concise review of zeolites, encompassing their definition, historical background, natural occurrence, geographical distribution, and diverse applications. Natural zeolites form over thousands of years when volcanic ash and rock react with alkaline groundwater. this yields over 40 distinct frameworks, though they are rarely found in a pure state and often contain other minerals or quartz as impurities. Zeolites, particularly sodium zeolites, are used to soften hard water by exchanging calcium and magnesium ions with sodium ions. they are also employed in water treatment for the removal of heavy metals and ammonia.

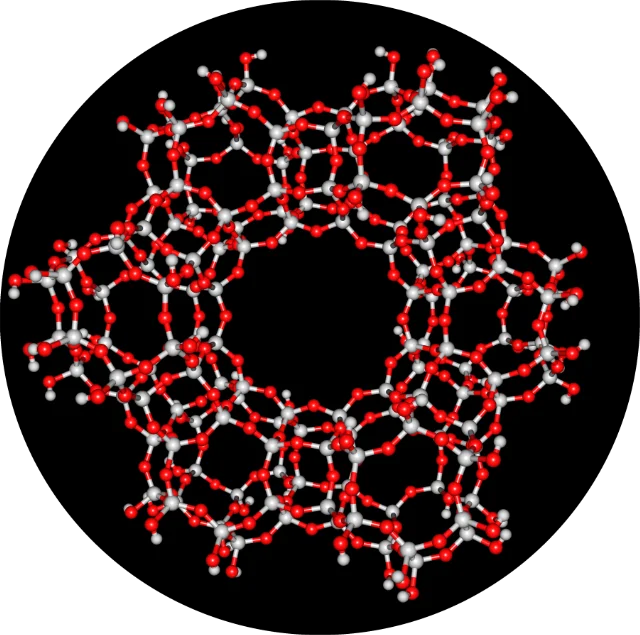
Zeolites Structure Properties And Uses Natural zeolites form over thousands of years when volcanic ash and rock react with alkaline groundwater. this yields over 40 distinct frameworks, though they are rarely found in a pure state and often contain other minerals or quartz as impurities. Zeolites, particularly sodium zeolites, are used to soften hard water by exchanging calcium and magnesium ions with sodium ions. they are also employed in water treatment for the removal of heavy metals and ammonia. Zeolites are crystalline aluminosilicates used as molecular sieves and ion exchangers. learn their structure, types, properties and uses. Zeolites are white solids with ordinary handling properties, like many routine aluminosilicate minerals, e.g. feldspar. they have the general formula where m is usually h and na . the si al ratio is variable, which provides a means to tune the properties. Zeolites are white solids with ordinary handling properties, like many routine aluminosilicate minerals, e.g. feldspar. they have the general formula (malo2) (sio2)x(h2o)y where m is usually h and na . the si al ratio is variable, which provides a means to tune the properties. Explore zeolite’s definition, structure, properties, and uses. master chemistry concepts easily with vedantu’s expert guidance.

Zeolites Structure Properties And Uses Zeolites are crystalline aluminosilicates used as molecular sieves and ion exchangers. learn their structure, types, properties and uses. Zeolites are white solids with ordinary handling properties, like many routine aluminosilicate minerals, e.g. feldspar. they have the general formula where m is usually h and na . the si al ratio is variable, which provides a means to tune the properties. Zeolites are white solids with ordinary handling properties, like many routine aluminosilicate minerals, e.g. feldspar. they have the general formula (malo2) (sio2)x(h2o)y where m is usually h and na . the si al ratio is variable, which provides a means to tune the properties. Explore zeolite’s definition, structure, properties, and uses. master chemistry concepts easily with vedantu’s expert guidance.

Zeolites Structure Properties And Uses Zeolites are white solids with ordinary handling properties, like many routine aluminosilicate minerals, e.g. feldspar. they have the general formula (malo2) (sio2)x(h2o)y where m is usually h and na . the si al ratio is variable, which provides a means to tune the properties. Explore zeolite’s definition, structure, properties, and uses. master chemistry concepts easily with vedantu’s expert guidance.
Zeolites Creative Chemistry
Comments are closed.