Workshop 1 Risk Assessment Of Genotoxic Compounds In The Presence Of Data From Carcinogenicity

Risk Assessment Of Non Carcinogenic And Carcinogenic Compounds The symposium aims to address the challenges and future directions in the risk assessment of genotoxic substances. Due to complex pathways and many tumor types, it is difficult to get data and to interpret them. at the present time, probably the carcinogenicity study is still required.

Risk Assessment And Management Of Genotoxic Risk Assessment And Facts, challenges and open questions i. genotoxicity data are mainly interpreted in a qualitative way, typically resulting in a binary classification of chemical entities. The symposium aims to address the challenges and future directions in the risk assessment of genotoxic substances. it will bring together experts from different disciplines either from academia, industry or regulatory authorities worldwide to present and discuss their activities. “for mutagenicity and carcinogenicity, a non threshold assessment should be carried out if the active substance or substance of concern is genotoxic and carcinogenic”. (committee for medicinal products for human use (chmp), european medicines agency (emea), assessment and control of dna reactive (mutagenic) impurities in pharmaceuticals to limit potential carcinogenic risk m7 (r2) liquid chromatography–mass spectrometry method for determination of n methyl n nitrosopyridin 4 amine in a vonoprazan.

Pdf Toxicogenomics Data For Chemical Safety Assessment And “for mutagenicity and carcinogenicity, a non threshold assessment should be carried out if the active substance or substance of concern is genotoxic and carcinogenic”. (committee for medicinal products for human use (chmp), european medicines agency (emea), assessment and control of dna reactive (mutagenic) impurities in pharmaceuticals to limit potential carcinogenic risk m7 (r2) liquid chromatography–mass spectrometry method for determination of n methyl n nitrosopyridin 4 amine in a vonoprazan. Here, we review current opportunities, challenges and perspectives for a more quantitative approach to genotoxicity assessment. Available data are more consistent with an absence of a robust, clinically meaningful peripheral blood genotoxic signal during ssri therapy, while acknowledging scarce longitudinal follow up, heterogeneous endpoints, and incomplete control for disease state, lifestyle, and co medications. For a successful risk assessment, in particular of genotoxic chemicals, several conditions and models such as the margin of exposure (moe) approach or the threshold of toxicological concern (ttc) concept exist, which will be discussed. Inherited mutations hazard identification and characterization 4 4 1 linked to human diseases are compiled in the human gene mutation 2 database (hgdb, 2017). 3 the overview presented in this chapter focuses on the identification 4 of mutagens and genotoxic carcinogens, consistent with the who ipcs.

Indicators Of Genotoxic Risk Download Table Here, we review current opportunities, challenges and perspectives for a more quantitative approach to genotoxicity assessment. Available data are more consistent with an absence of a robust, clinically meaningful peripheral blood genotoxic signal during ssri therapy, while acknowledging scarce longitudinal follow up, heterogeneous endpoints, and incomplete control for disease state, lifestyle, and co medications. For a successful risk assessment, in particular of genotoxic chemicals, several conditions and models such as the margin of exposure (moe) approach or the threshold of toxicological concern (ttc) concept exist, which will be discussed. Inherited mutations hazard identification and characterization 4 4 1 linked to human diseases are compiled in the human gene mutation 2 database (hgdb, 2017). 3 the overview presented in this chapter focuses on the identification 4 of mutagens and genotoxic carcinogens, consistent with the who ipcs.

Pdf Genotoxic Impurities And Its Risk Assessment In Drug Compounds For a successful risk assessment, in particular of genotoxic chemicals, several conditions and models such as the margin of exposure (moe) approach or the threshold of toxicological concern (ttc) concept exist, which will be discussed. Inherited mutations hazard identification and characterization 4 4 1 linked to human diseases are compiled in the human gene mutation 2 database (hgdb, 2017). 3 the overview presented in this chapter focuses on the identification 4 of mutagens and genotoxic carcinogens, consistent with the who ipcs.
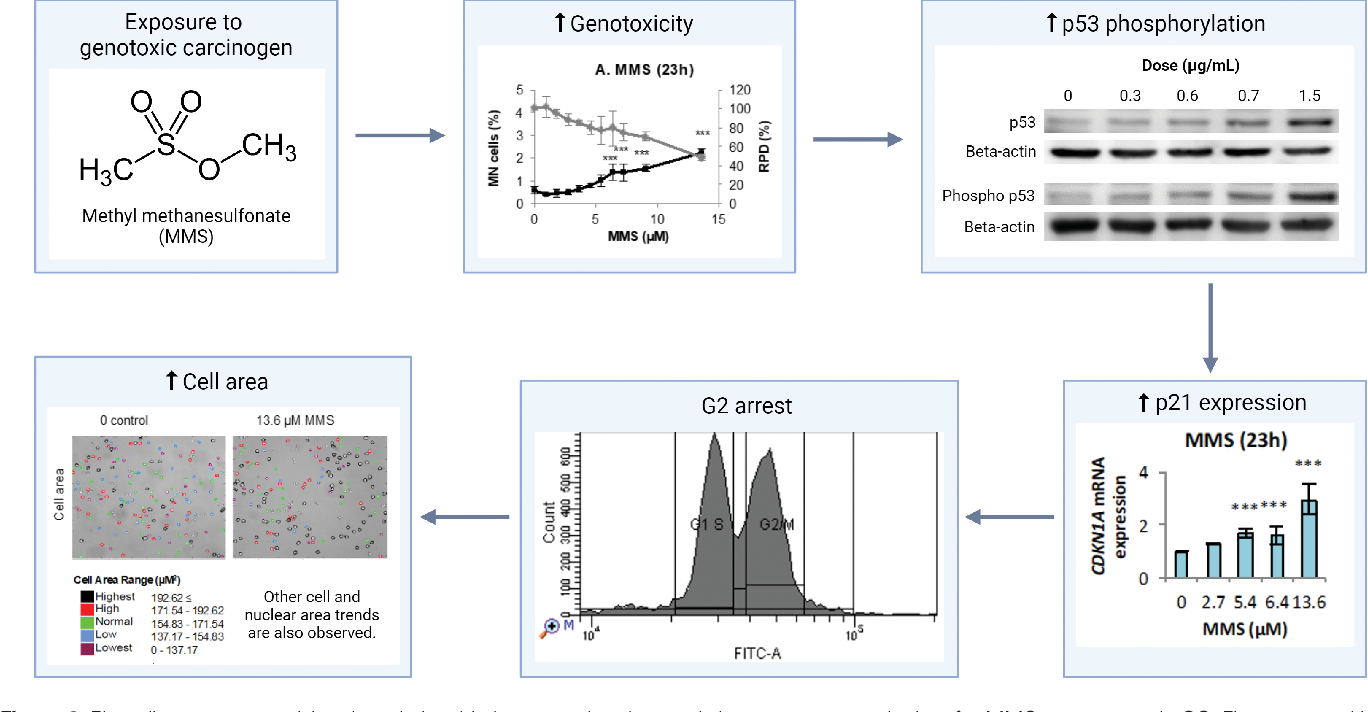
Figure 1 From An Integrated In Vitro Carcinogenicity Test That
Comments are closed.