Why Is Ice Slippery

Why Is Ice Slippery Era Observer The reason we can gracefully glide on an ice skating rink or clumsily slip on an icy sidewalk is that the surface of ice is coated by a thin watery layer. scientists generally agree that this lubricating, liquidlike layer is what makes ice slippery. Discover why ice is slippery through the lens of friction physics. this article breaks down the science explained behind ice’s slippery surface in simple, engaging terms.

Why Is Ice Slippery Mental Floss New research conducted at saarland university reveals that it's not pressure or friction that causes ice to become slippery, but rather the interaction between molecular dipoles in the ice and. Everyday experience suggests why ice surfaces should be slippery: water spilled on a kitchen floor or rainwater on asphalt or concrete can create the same kinds of hazards for walkers and drivers that ice can. Slippery ice is something most of us have encountered, whether it’s been under our boots, car tires or skates. but why ice is so slick has puzzled scientists for centuries. now researchers in germany offer a new explanation. they show ice can melt slightly with no heat added. For nearly two centuries, scientists believed that ice becomes slippery because pressure or friction melts its surface. new research from saarland university overturns this idea. for more than a century, students around the world have been told that pressure and friction make ice melt.

Why Is Ice Slippery Slippery ice is something most of us have encountered, whether it’s been under our boots, car tires or skates. but why ice is so slick has puzzled scientists for centuries. now researchers in germany offer a new explanation. they show ice can melt slightly with no heat added. For nearly two centuries, scientists believed that ice becomes slippery because pressure or friction melts its surface. new research from saarland university overturns this idea. for more than a century, students around the world have been told that pressure and friction make ice melt. Because ice is less dense than liquid water, its melting point is lowered under high pressures. a long standing theory says that this is what causes ice to be slippery: as you step on it, the. Learn from a scientist why ice is slippery and how it affects winter driving. watch a video and explore a model of a car sliding on ice. Ice’s melty coating appears even at temperatures well below freezing, a phenomenon known as “premelting.” that layer acts as a lubricant, explaining why ice is slippery even under frigid. Scientists discovered that ice is slippery not from pressure or heat, but from molecular dipoles disrupting its crystal surface.
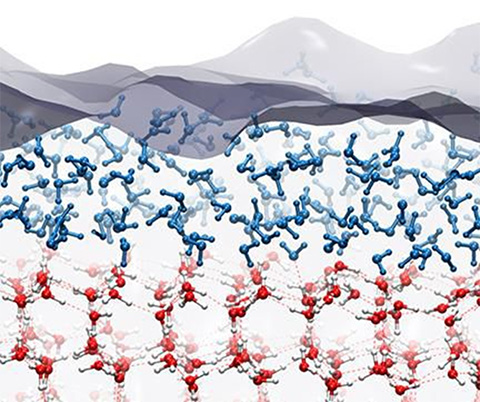
Why Is Ice Slippery Earthdate Because ice is less dense than liquid water, its melting point is lowered under high pressures. a long standing theory says that this is what causes ice to be slippery: as you step on it, the. Learn from a scientist why ice is slippery and how it affects winter driving. watch a video and explore a model of a car sliding on ice. Ice’s melty coating appears even at temperatures well below freezing, a phenomenon known as “premelting.” that layer acts as a lubricant, explaining why ice is slippery even under frigid. Scientists discovered that ice is slippery not from pressure or heat, but from molecular dipoles disrupting its crystal surface.

German Scientists Debunk 200 Year Old Theory On Why Ice Is Slippery Ice’s melty coating appears even at temperatures well below freezing, a phenomenon known as “premelting.” that layer acts as a lubricant, explaining why ice is slippery even under frigid. Scientists discovered that ice is slippery not from pressure or heat, but from molecular dipoles disrupting its crystal surface.
Comments are closed.