What Is The Difference Between Mapping And Validation

What Is The Difference Between Mapping And Validation Explore the differences between “validation” and “mapping” and why they are each important. Ever wondered what a validation, qualification, or temperature mapping means and how it is applied in the gxp regulated environment? this article answers these questions while explaining how each process supports regulatory compliance, product quality, and patient safety.

What Is The Difference Between Mapping And Validation A mapping study defines the behavioral pattern within the room, while the qualification study follows through with a detailed analysis to show the design and operations match the expected results. Both are crucial in fda regulated industries. however, mapping is more about environmental consistency and storage conditions, while thermal validation is process based and time sensitive. understanding the difference helps organizations apply the right controls and avoid costly compliance gaps. Generally, while the initial qualification will usually include mapping of the empty chamber and the loaded chamber, subsequent re qualifications only require mapping of the loaded chamber. Verifying that equipment and storage facilities meet basic temperature requirements is a fundamental component of a thermal mapping process. by providing evidence of the temperature within a controlled area, the overall process begins on stable ground.

What Is The Difference Between Temperature Mapping And Validation Generally, while the initial qualification will usually include mapping of the empty chamber and the loaded chamber, subsequent re qualifications only require mapping of the loaded chamber. Verifying that equipment and storage facilities meet basic temperature requirements is a fundamental component of a thermal mapping process. by providing evidence of the temperature within a controlled area, the overall process begins on stable ground. By understanding the differences between validation and qualification and the role of thermal mapping, organizations can ensure the quality and safety of their products and meet regulatory requirements. key takeaways: qualification focuses on equipment, while validation focuses on the entire process. Temperature mapping study and validation are mainly applicable to the pharmaceutical industry and food fmcg industry. it helps us to analyze and ensure the uniformity of temperature distribution across all areas of the controlled areas under real life conditions. In the methods section, we describe how we reviewed the use of the words validity and validation in research methodology and modelling textbooks, and how we synthesized both concepts with the adapted modelling cycle into the ‘map of validity and validation in cas modelling’. In practice, most pharmaceutical companies use temperature mapping for cold storage qualification, but thermal mapping may be applied in larger or more complex facilities where multiple environmental factors must be validated.
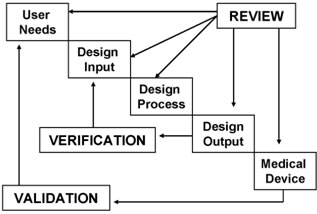
Difference Between Verification And Validation Verification Vs Validation By understanding the differences between validation and qualification and the role of thermal mapping, organizations can ensure the quality and safety of their products and meet regulatory requirements. key takeaways: qualification focuses on equipment, while validation focuses on the entire process. Temperature mapping study and validation are mainly applicable to the pharmaceutical industry and food fmcg industry. it helps us to analyze and ensure the uniformity of temperature distribution across all areas of the controlled areas under real life conditions. In the methods section, we describe how we reviewed the use of the words validity and validation in research methodology and modelling textbooks, and how we synthesized both concepts with the adapted modelling cycle into the ‘map of validity and validation in cas modelling’. In practice, most pharmaceutical companies use temperature mapping for cold storage qualification, but thermal mapping may be applied in larger or more complex facilities where multiple environmental factors must be validated.

Mapping Validation Industry Degreetek In the methods section, we describe how we reviewed the use of the words validity and validation in research methodology and modelling textbooks, and how we synthesized both concepts with the adapted modelling cycle into the ‘map of validity and validation in cas modelling’. In practice, most pharmaceutical companies use temperature mapping for cold storage qualification, but thermal mapping may be applied in larger or more complex facilities where multiple environmental factors must be validated.
Comments are closed.