Vapor Pressure
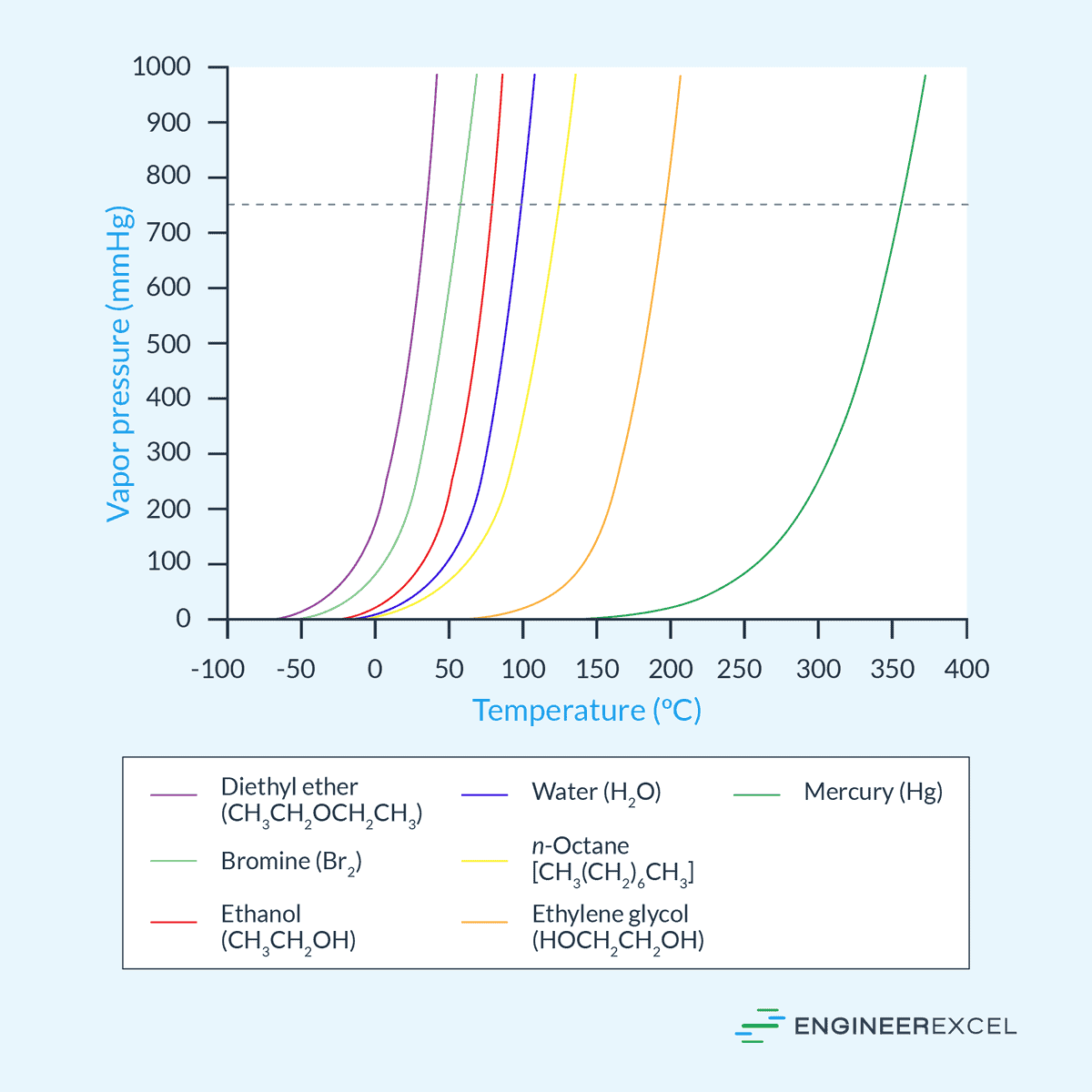
Vapor Pressure A Comprehensive Overview Engineerexcel Vapor pressure is the pressure exerted by a vapor in equilibrium with its condensed phases at a given temperature. learn how to measure vapor pressure, how it relates to boiling point and phase diagrams, and how it affects volatile substances and mixtures. Learn what vapor pressure is, how it depends on temperature and intermolecular forces, and how to calculate it using raoult's law or the clausius clapeyron equation. see examples, graphs, and tables of vapor pressure values for various substances.
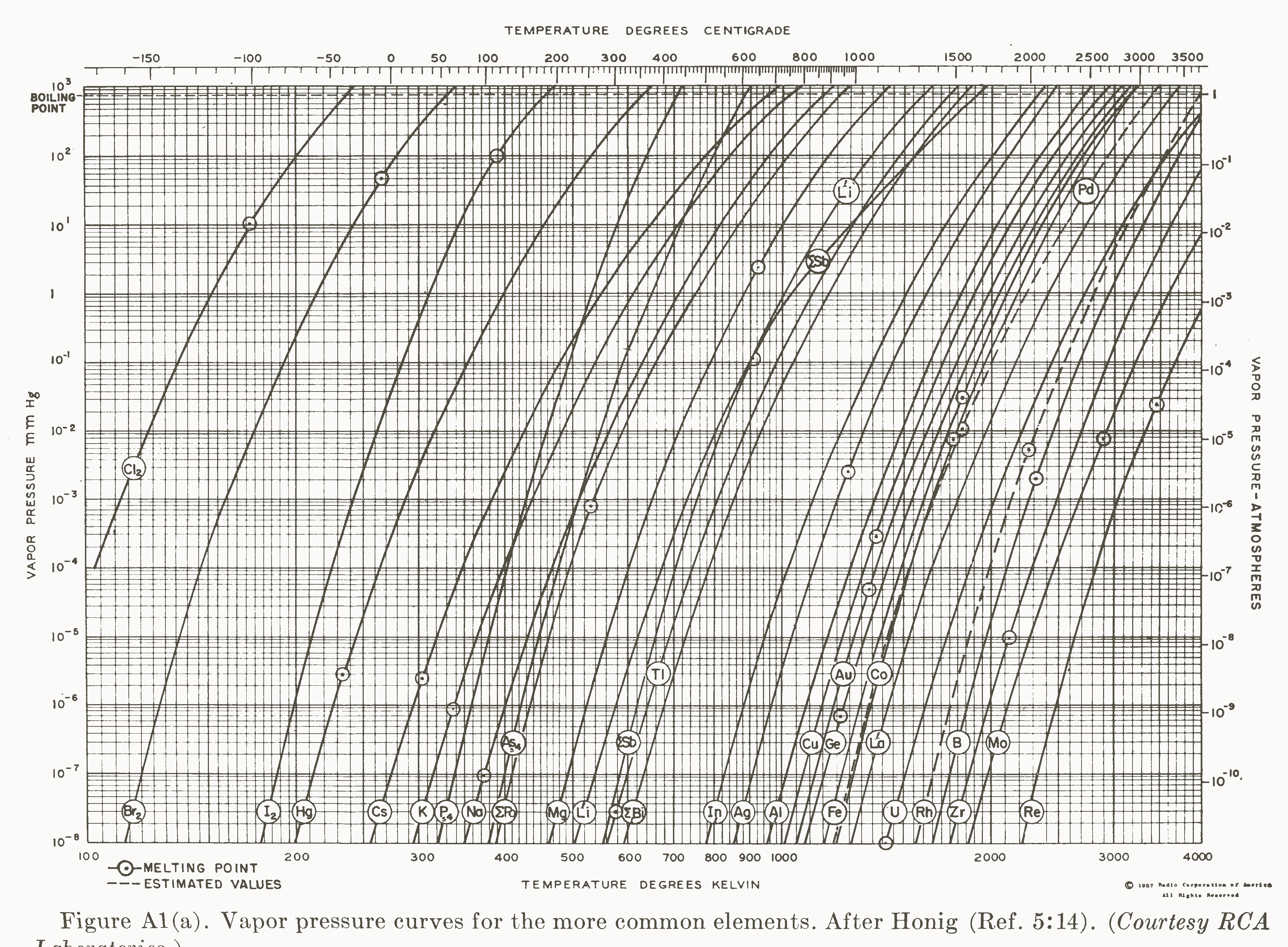
Vapor Pressures Of The Chemical Elements Vapor Pressure Of Metals And Vapor pressure is the pressure exerted by a gas when it is in equilibrium with its liquid or solid form. learn how to calculate it using raoult's law and the clausius clapeyron equation, and see how it depends on temperature and the nature of the liquid. In this section, we describe vapor pressure in more detail and explain how to quantitatively determine the vapor pressure of a liquid. Learn what vapor pressure is and how it depends on temperature for different liquids. find approximate vapor pressure values for common fluids at atmospheric pressure and 20 25 oc range. Vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phase (liquid or solid) at a given temperature in a closed system. it indicates the evaporation rate of the liquid and the tendency of particles to escape into the vapor phase.
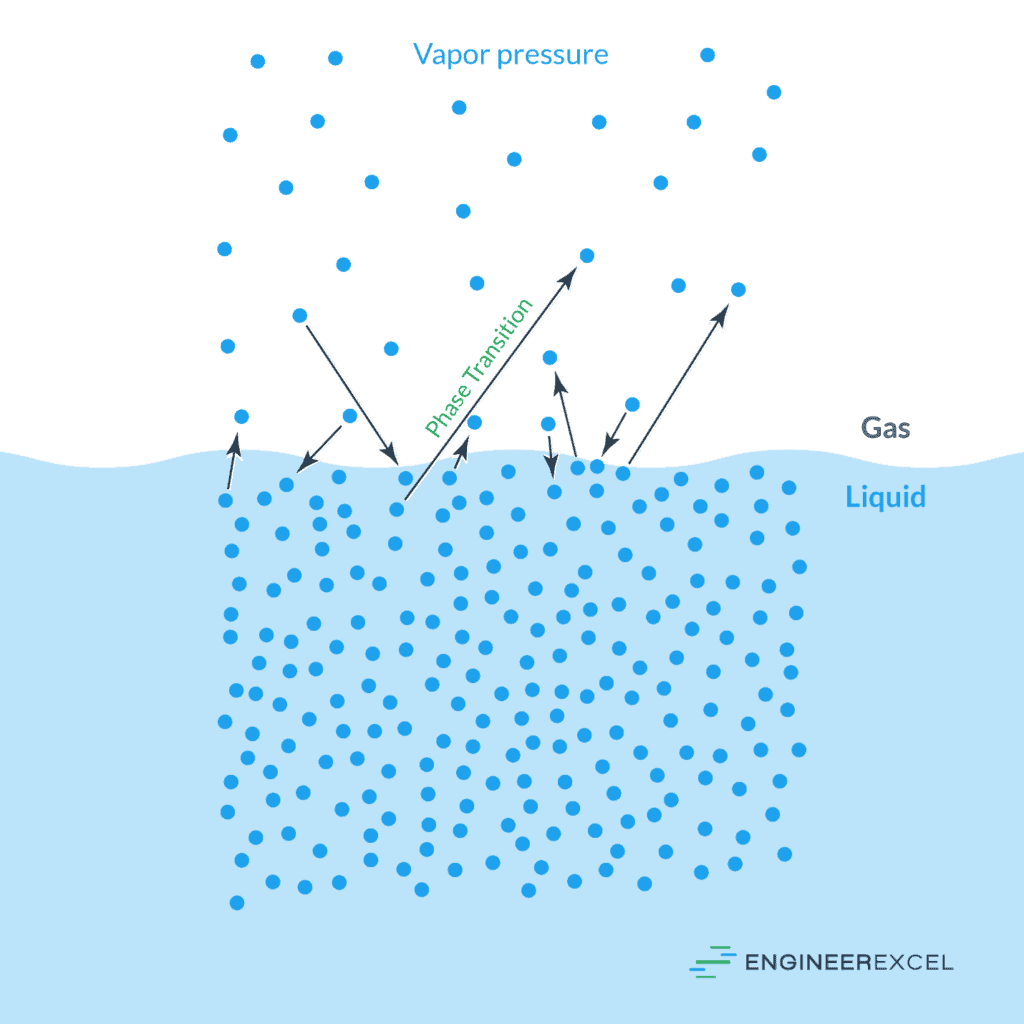
Vapor Pressure Of Water Explained Engineerexcel Learn what vapor pressure is and how it depends on temperature for different liquids. find approximate vapor pressure values for common fluids at atmospheric pressure and 20 25 oc range. Vapor pressure is defined as the pressure exerted by a vapor in thermodynamic equilibrium with its condensed phase (liquid or solid) at a given temperature in a closed system. it indicates the evaporation rate of the liquid and the tendency of particles to escape into the vapor phase. Learn what vapor pressure is, how it varies with temperature and types of molecules, and how it is measured. see examples of vapor pressure for different substances and graphs of vapor pressure vs. temperature. Learn what vapor pressure is, how it depends on temperature and intermolecular forces, and how to calculate it using equations. explore the practical applications of vapor pressure in distillation, refrigeration, and relief devices. Vapor pressure is a measure of how eagerly molecules escape from a liquid (or solid) into the gas phase. when evaporation and condensation balance out, the vapor pressure represents a dynamic equilibrium. Vapour pressure, pressure exerted by a vapour when the vapour is in equilibrium with the liquid or solid form, or both, of the same substance—i.e., when conditions are such that the substance can exist in both or in all three phases. learn more about vapour pressure in this article.
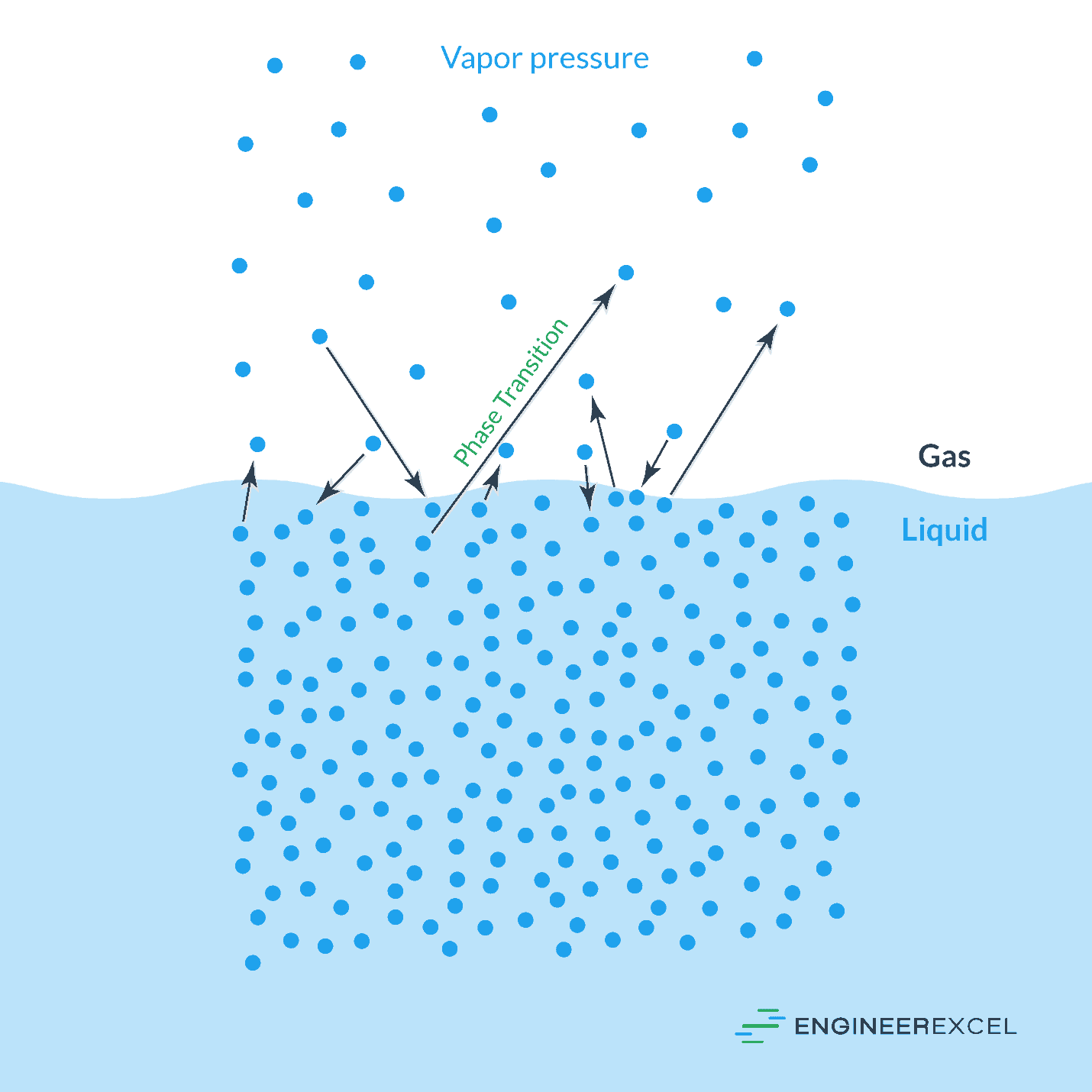
Vapor Pressure Of Water Explained Engineerexcel Learn what vapor pressure is, how it varies with temperature and types of molecules, and how it is measured. see examples of vapor pressure for different substances and graphs of vapor pressure vs. temperature. Learn what vapor pressure is, how it depends on temperature and intermolecular forces, and how to calculate it using equations. explore the practical applications of vapor pressure in distillation, refrigeration, and relief devices. Vapor pressure is a measure of how eagerly molecules escape from a liquid (or solid) into the gas phase. when evaporation and condensation balance out, the vapor pressure represents a dynamic equilibrium. Vapour pressure, pressure exerted by a vapour when the vapour is in equilibrium with the liquid or solid form, or both, of the same substance—i.e., when conditions are such that the substance can exist in both or in all three phases. learn more about vapour pressure in this article.
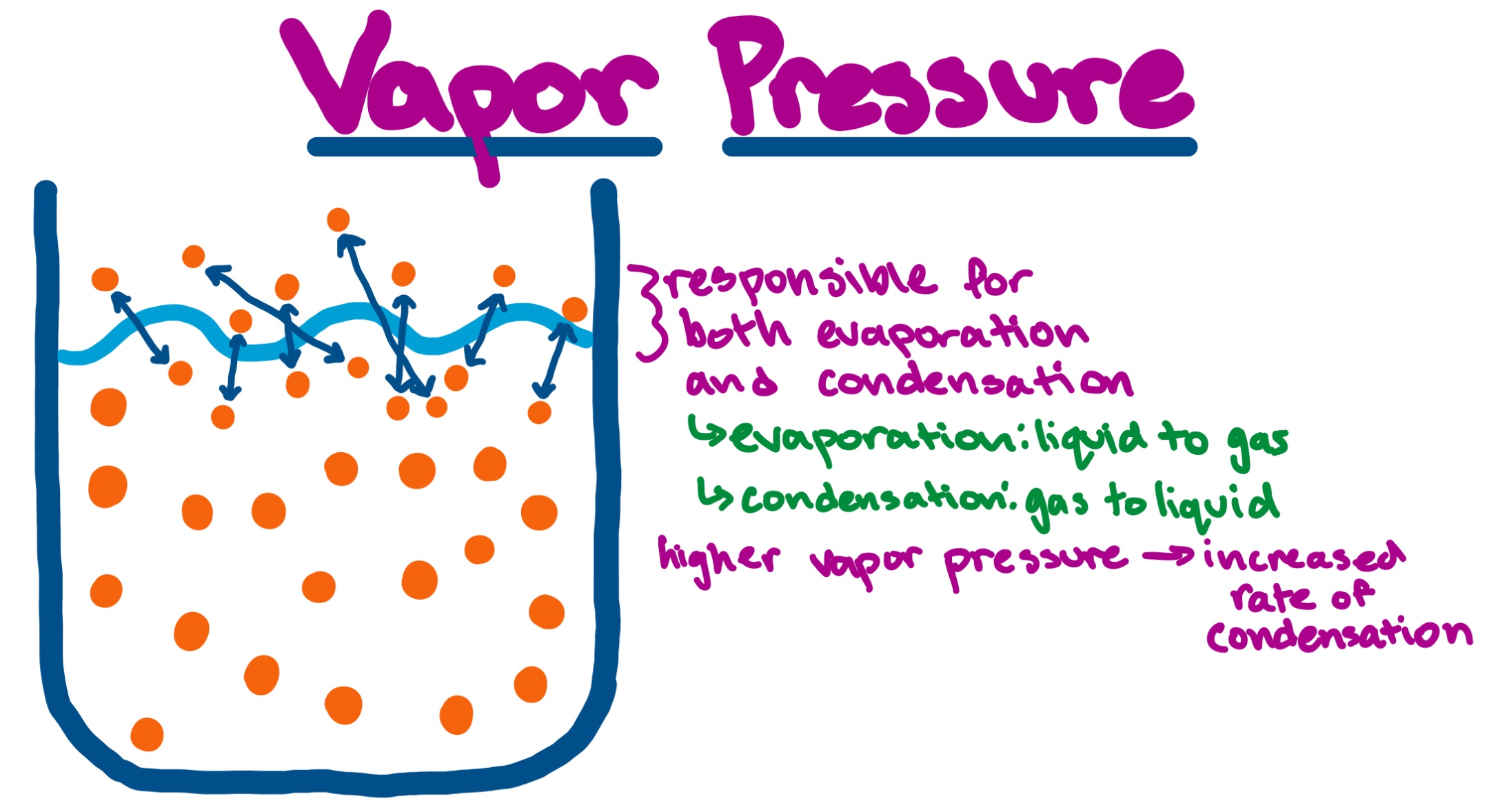
Vapor Pressure Definition Overview Expii Vapor pressure is a measure of how eagerly molecules escape from a liquid (or solid) into the gas phase. when evaporation and condensation balance out, the vapor pressure represents a dynamic equilibrium. Vapour pressure, pressure exerted by a vapour when the vapour is in equilibrium with the liquid or solid form, or both, of the same substance—i.e., when conditions are such that the substance can exist in both or in all three phases. learn more about vapour pressure in this article.
Comments are closed.