Validation Process And Equipment

Equipment Validation Presentationeze Discover the basics of equipment validation in manufacturing: its importance, regulatory standards, and steps to compliance and quality. What is equipment validation? equipment validation is a documented process demonstrating whether a piece of equipment, system, or process consistently produces results within predefined specifications.

Equipment Validation Presentationeze Equipment validation is a systematic and documented process that provides confidence in the reliability, consistency, and quality of manufacturing processes. it is a proactive approach to quality assurance, ensuring that products meet regulatory requirements and customer expectations. Equipment validation is the process of validating the requirements, specifications, and uses of a piece of equipment to ensure it meets user needs as well as various regulatory and safety requirements. This guidance incorporates principles and approaches that all manufacturers can use to validate manufacturing processes. All processes and systems once validated will be maintained in a validated state through the life cycle of the process system. maintaining the validated state will be achieved by the change control system, re qualification, training, sops, calibration and engineering maintenance programmes.

Equipment Validation Facility Qualification Material This guidance incorporates principles and approaches that all manufacturers can use to validate manufacturing processes. All processes and systems once validated will be maintained in a validated state through the life cycle of the process system. maintaining the validated state will be achieved by the change control system, re qualification, training, sops, calibration and engineering maintenance programmes. Equipment validation is a systematic, documented process used in highly regulated manufacturing environments. it provides comprehensive proof that a piece of equipment functions reliably and consistently for its specific intended use. Ensuring laboratory equipment operates as expected is critically important to the validity of results generated by the laboratory. as part of process validation through iq oq pq, laboratories verify and validate that equipment performs to pre determined specifications or criteria. Learn how to implement rigorous process validation techniques in regulated industries. process validation isn’t just about checking boxes for regulators—it’s about building quality into products from the ground up. As a component of quality assurance, equipment or machine validation is absolutely critical to produce consistent, high quality products. the below checklist can help manufacturers quickly develop their validation plan and at the same time it will help the validation procedure.

Equipment Validation Pharma4ever Equipment validation is a systematic, documented process used in highly regulated manufacturing environments. it provides comprehensive proof that a piece of equipment functions reliably and consistently for its specific intended use. Ensuring laboratory equipment operates as expected is critically important to the validity of results generated by the laboratory. as part of process validation through iq oq pq, laboratories verify and validate that equipment performs to pre determined specifications or criteria. Learn how to implement rigorous process validation techniques in regulated industries. process validation isn’t just about checking boxes for regulators—it’s about building quality into products from the ground up. As a component of quality assurance, equipment or machine validation is absolutely critical to produce consistent, high quality products. the below checklist can help manufacturers quickly develop their validation plan and at the same time it will help the validation procedure.
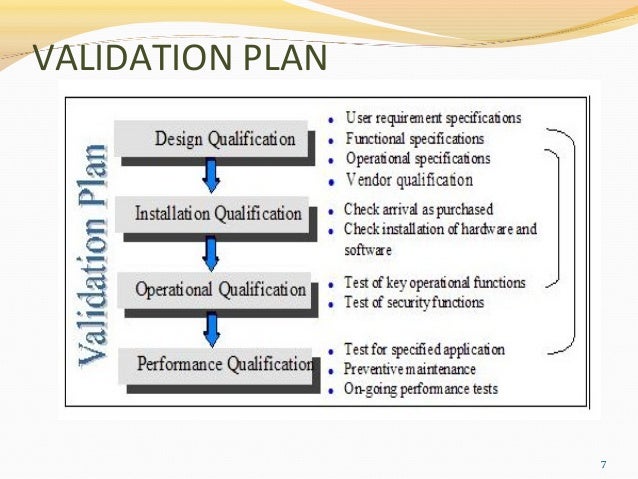
Equipment Validation Process Guideline For Equipment And System Learn how to implement rigorous process validation techniques in regulated industries. process validation isn’t just about checking boxes for regulators—it’s about building quality into products from the ground up. As a component of quality assurance, equipment or machine validation is absolutely critical to produce consistent, high quality products. the below checklist can help manufacturers quickly develop their validation plan and at the same time it will help the validation procedure.

Process Validation Procedure 4easyreg
Comments are closed.