Uv Light Fluorescence Spectrum
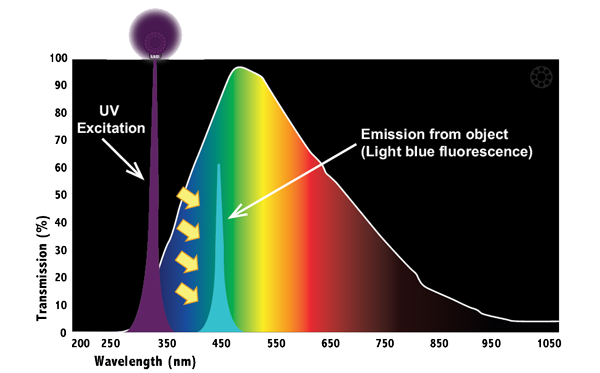
Uv Light Fluorescence Spectrum It is widely applied in chemistry, biochemistry, and environmental science for compound identification and quantification. this review examines the principles of uv absorption, the types of uv spectrometers, and the integration of uv spectroscopy with other analytical methods. However, it can be beneficial and informative to obtain the entire fluorescence landscape (also known as 2d fluorescence spectroscopy) in order to find the exact excitation and emission maxima as well as the correct structure of the peaks.

Uv Light Fluorescence Spectrum It involves using a beam of light, usually ultraviolet light, that excites the electrons in molecules of certain compounds and causes them to emit light; typically, but not necessarily, visible light. The sample gas is irradiated with ultraviolet light of a specific wavelength while flowing through a fluorescence cell. depending on the so 2 concentration in the sample gas, the uv light absorption changes, and the intensity of light from fluorescence also changes. Fluorescence techniques are nondestructive analytical methods used in a wide range of applications. since many fluorophores of interest can be excited with uv light and nowadays compact solid state uv emitters are available, uv fluorescence methods are emerging. In this two part series, we will describe both uv vis and fluorescence spectroscopy by comparing their characteristics, such as their operating principle, system composition, and advantages and disadvantages.
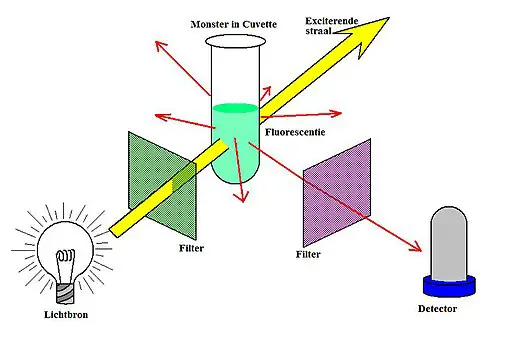
Uv Light Fluorescence Spectrum Fluorescence techniques are nondestructive analytical methods used in a wide range of applications. since many fluorophores of interest can be excited with uv light and nowadays compact solid state uv emitters are available, uv fluorescence methods are emerging. In this two part series, we will describe both uv vis and fluorescence spectroscopy by comparing their characteristics, such as their operating principle, system composition, and advantages and disadvantages. Once the sample is loaded into the cell, a collimated (almost parallel) uv light source passes through the sample so that it will fluoresce. a monochromator is often used, either between the light source and the sample, or between the sample and the detector. Learn the basic properties of the light spectrum and its importance in fluorescence. In fluorescence spectroscopy, high energy light from an excitation source, such as uv light source or laser, is directed towards your sample. the sample absorbs this light, exciting all available electrons into their higher energy state. Ultraviolet (uv) fluorescence describes a process where certain substances absorb invisible ultraviolet light and then immediately release that energy as visible light. this transformation reveals a hidden world of vibrant colors not apparent under normal lighting conditions.
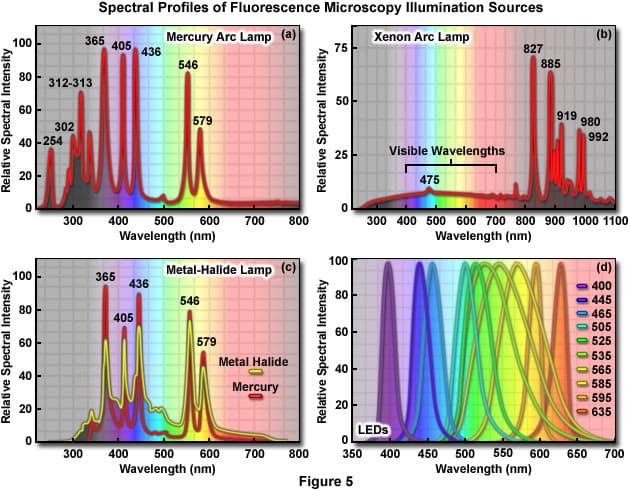
Uv Light Fluorescence Spectrum Once the sample is loaded into the cell, a collimated (almost parallel) uv light source passes through the sample so that it will fluoresce. a monochromator is often used, either between the light source and the sample, or between the sample and the detector. Learn the basic properties of the light spectrum and its importance in fluorescence. In fluorescence spectroscopy, high energy light from an excitation source, such as uv light source or laser, is directed towards your sample. the sample absorbs this light, exciting all available electrons into their higher energy state. Ultraviolet (uv) fluorescence describes a process where certain substances absorb invisible ultraviolet light and then immediately release that energy as visible light. this transformation reveals a hidden world of vibrant colors not apparent under normal lighting conditions.
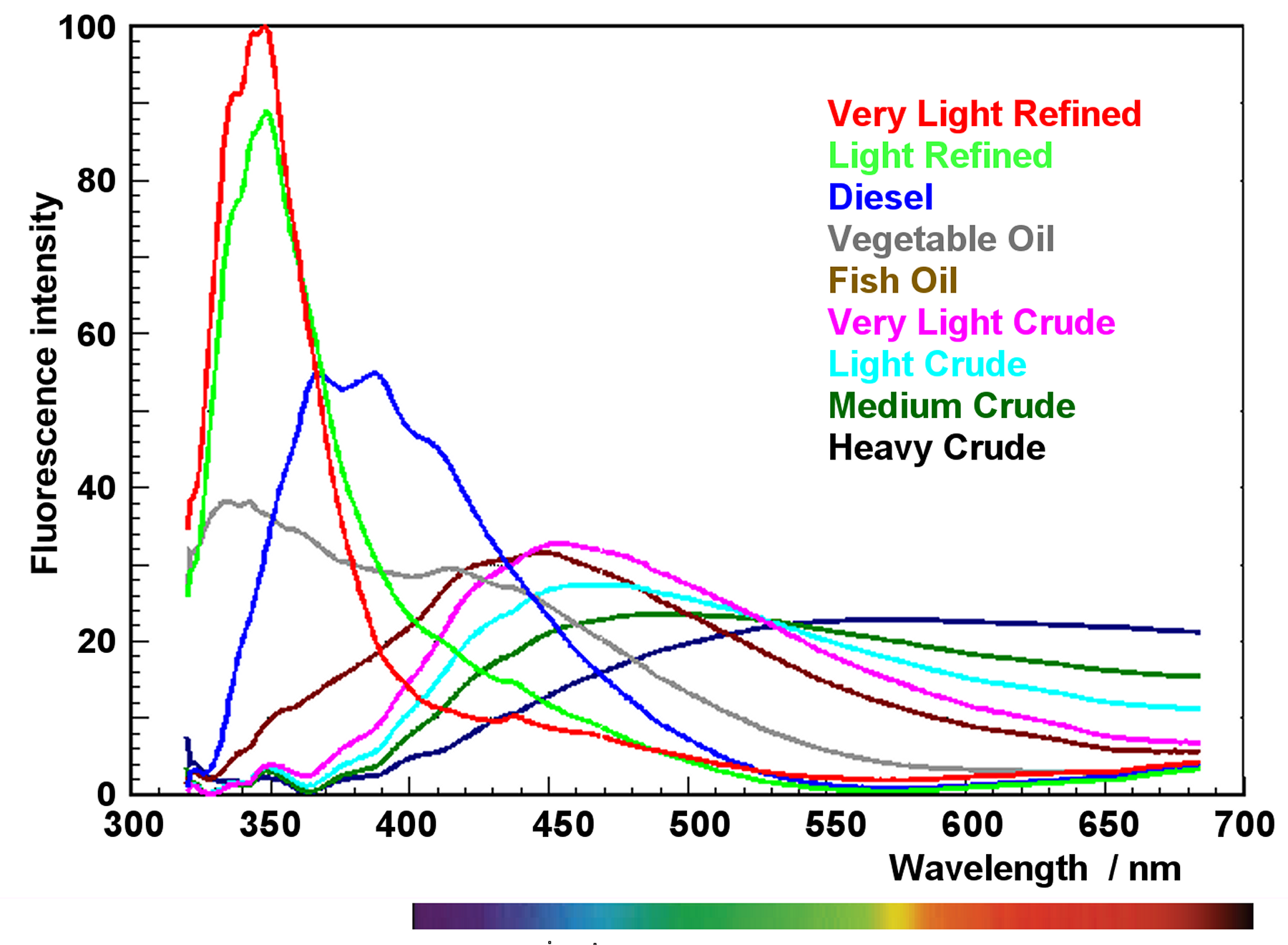
Uv Light Fluorescence Spectrum In fluorescence spectroscopy, high energy light from an excitation source, such as uv light source or laser, is directed towards your sample. the sample absorbs this light, exciting all available electrons into their higher energy state. Ultraviolet (uv) fluorescence describes a process where certain substances absorb invisible ultraviolet light and then immediately release that energy as visible light. this transformation reveals a hidden world of vibrant colors not apparent under normal lighting conditions.
Comments are closed.