Unit 1 Atomic Structure Nuclear Chemistry
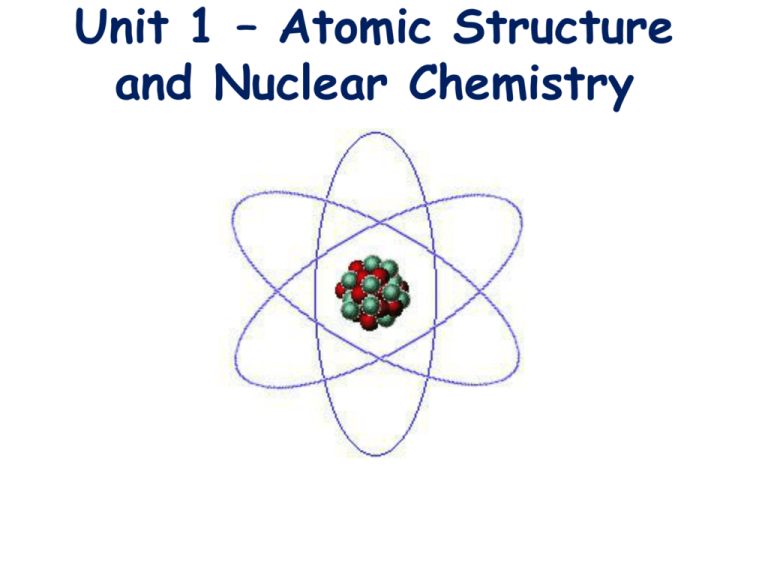
Unit 1 Atomic Structure And Nuclear Chemistry Nucleus (protons and neutrons) is small and dense and contains most of the mass of the atom. the electron cloud (where electrons are found) contain most of the volume (3 d space) of an atom. This document provides an overview of atomic structure and nuclear chemistry concepts. it defines the atom and its components, including electrons, protons, and neutrons.
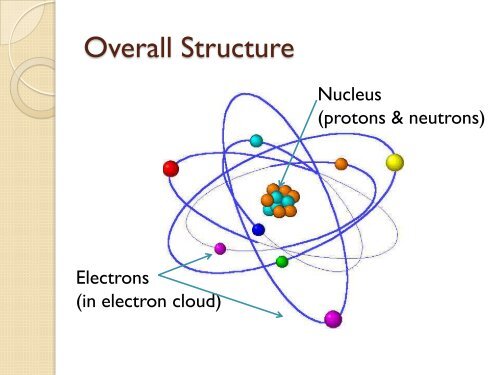
Structure Of An Atom Use the principles of atomic structure and or chemical bonding to explain each of the following. in each part, your answers must include references to both substances. Comprehensive lecture notes on atomic structure, isotopes, radioactivity, nuclear reactions, fission, and fusion. high school chemistry. The kanji characters for "atom." this image was formed by using the tiny tip of an stm to pick up individual atoms of iron and place them on a copper (111) surface. Unit 1 in class review.

Unit 1 Atomic Structure Nuclear Chemistry The kanji characters for "atom." this image was formed by using the tiny tip of an stm to pick up individual atoms of iron and place them on a copper (111) surface. Unit 1 in class review. Unit 1 atomic structure notes free download as pdf file (.pdf) or read online for free. the document discusses atomic structure, focusing on electron energy levels and their behavior in atoms. In 1911, ernest rutherford introduced the nuclear model, showing that most of the atom’s mass and positive charge is concentrated in a tiny nucleus, with electrons revolving around it. in 1913, niels bohr improved this model by proposing quantized energy levels (shells) for electrons. It is what holds atomic nuclei together and confines quarks in nuclear particles such as protons and neutrons. again, like gravitation, there is very little in chemistry where we need to consider the strong and weak forces in our analysis except for nuclear reactions. Study with quizlet and memorize flashcards containing terms like what is matter, what is mass volume density, what determines what phase a substance is in and more.

Unit 1 Atomic Structure Objectives Ppt Download Unit 1 atomic structure notes free download as pdf file (.pdf) or read online for free. the document discusses atomic structure, focusing on electron energy levels and their behavior in atoms. In 1911, ernest rutherford introduced the nuclear model, showing that most of the atom’s mass and positive charge is concentrated in a tiny nucleus, with electrons revolving around it. in 1913, niels bohr improved this model by proposing quantized energy levels (shells) for electrons. It is what holds atomic nuclei together and confines quarks in nuclear particles such as protons and neutrons. again, like gravitation, there is very little in chemistry where we need to consider the strong and weak forces in our analysis except for nuclear reactions. Study with quizlet and memorize flashcards containing terms like what is matter, what is mass volume density, what determines what phase a substance is in and more.

Fa1 For Unit 1 Atomic Structure Pdf It is what holds atomic nuclei together and confines quarks in nuclear particles such as protons and neutrons. again, like gravitation, there is very little in chemistry where we need to consider the strong and weak forces in our analysis except for nuclear reactions. Study with quizlet and memorize flashcards containing terms like what is matter, what is mass volume density, what determines what phase a substance is in and more.
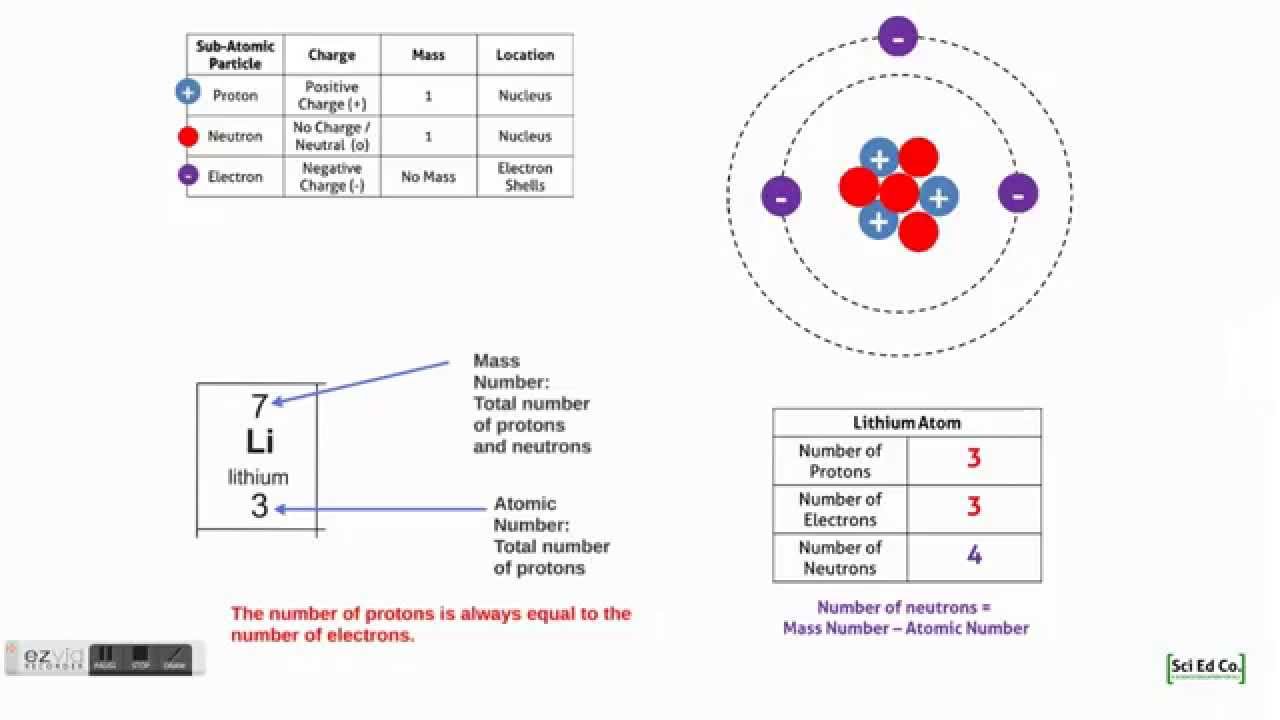
Sci Ed Co Gcse Aqa Chemistry Unit 1 Atoms And Atomic Structure
Comments are closed.