Types Of Solutions
Types Of Solutions Different Types Homogeneous Heterogeneous In all solutions, whether gaseous, liquid, or solid, the substance present in the greatest amount is the solvent, and the substance or substances present in lesser amounts are the solute (s). A solution is a type of mixture in which one substance, the solute, is completely dissolved in another substance, the solvent. learn about the different types of solutions based on the physical state of the solute and solvent and the amount of solute dissolved.
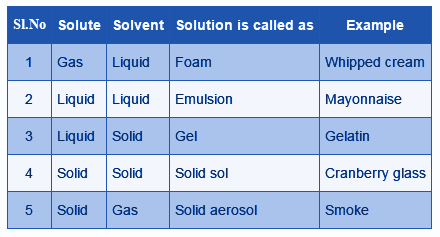
Types Of Solutions Different Types Homogeneous Heterogeneous The most common type of solution is the liquid solution (gas in liquid, liquid in liquid, solid in liquid). in all, we can divide solutions into nine different classes as follows:. Solutions can be classified as dilute solutions when the proportion of solute to solvent is low, and concentrated solutions when the proportion of solute to solvent is high. On the basis of physical states of solvent and solute can be categorized as solid, liquid and gaseous solutions. in solid solutions, solute and solvent are in the solid state. for example ceramics and polymer blends. in liquid solutions, solid, gas or liquid is mixed in a liquid state. Solutions are primarily classified according to the physical states of solute and solvent—solid, liquid, or gas—producing nine possible types. in every case, the phase of the solvent decides the phase of the overall solution.

Types Of Solutions Different Types Homogeneous Heterogeneous On the basis of physical states of solvent and solute can be categorized as solid, liquid and gaseous solutions. in solid solutions, solute and solvent are in the solid state. for example ceramics and polymer blends. in liquid solutions, solid, gas or liquid is mixed in a liquid state. Solutions are primarily classified according to the physical states of solute and solvent—solid, liquid, or gas—producing nine possible types. in every case, the phase of the solvent decides the phase of the overall solution. Knowing the types of solutions helps in understanding chemical properties and reactions effectively. whether it’s a gas in gas or solid in liquid, every type plays a crucial role in chemistry and industry. Learn how solutions form from homogeneous mixtures of solvents and solutes, and how they exhibit different properties depending on their components and interactions. explore examples of solutions of different phases, such as gases, liquids, and solids. Solutions are combinations of different solutes and a solvent. filtration cannot separate the constituents of a solute. saturated, unsaturated, and supersaturated solutions are all possible. When the solvent is in solid state, solution is called solid solution. when the solvent is in liquid state, solution is called liquid solution. when the solvent is in gaseous state, solution is called gaseous solution.
Comments are closed.