Trends In The Properties Of Group 13 Elements The P Block Elements Chemistry Khan Academy
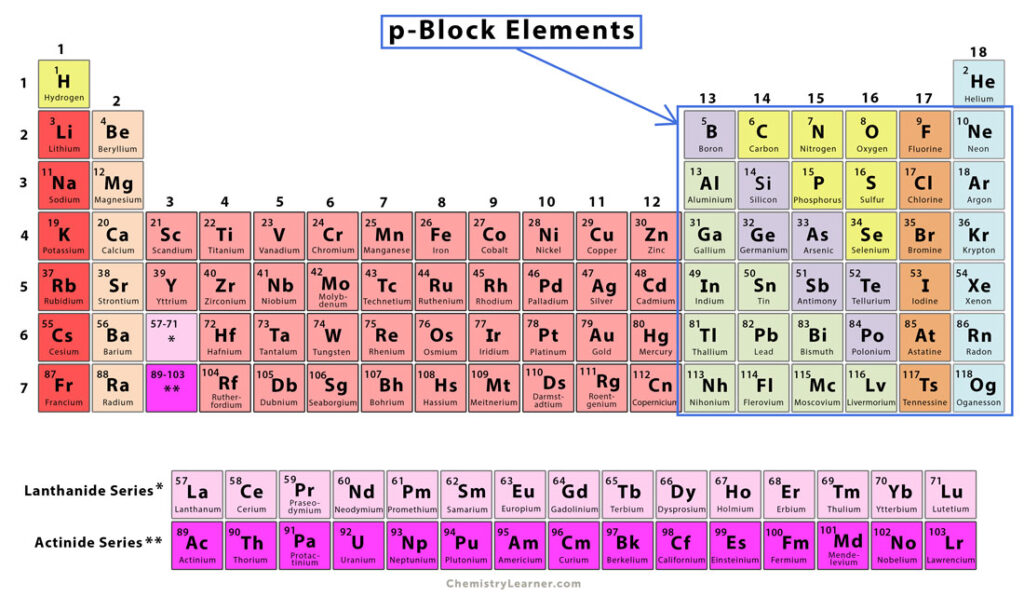
P Block Elements Chemistry Learner In this video, we will look at the various properties of the group 13 elements like atomic radius, density, ionisation enthalpy, reducing property and electronegativity and find out how and why they vary within the group. 00:15 electronic configurations of the group 13 elements. 00:56 is boron a non metal or a metalloid? 01:51 poor. In this video, we will look at the various properties of the group 13 elements like atomic radius, density, ionisation enthalpy, reducing property and electronegativity and find out how and.

Iit Jee Neet Learn Mastering P Block Elements Chemistry Full Course Level up on all the skills in this unit and collect up to 1,800 mastery points!. Unit 8: the p block elements group 13 elements learn introduction to p block elements. Through this exercise, we will test our understanding of the trends in the physical and chemical properties of group 13 elements. Group 13 elements learn introduction to p block elements oxidation states of p block elements.

Group 13 P Block Elements Periodic Physical Chemical Properties Through this exercise, we will test our understanding of the trends in the physical and chemical properties of group 13 elements. Group 13 elements learn introduction to p block elements oxidation states of p block elements. These elements are found in group 13 (xiii) of the p block in the periodic table of elements. aluminum, gallium, indium, and thallium are metallic. they each have three electrons in their outermost shell (a full s orbital and one electron in the p orbital) with the valence electron configuration ns 2 np 1. The document discusses the physical and chemical properties of group 13 elements, including trends in atomic and ionic radii, ionization energy, electronegativity, density, melting points, and metallic character. Learn group 13 elements, their properties, periodic trends, and memory tips for exams. essential for jee, neet, and board preparation. In p block, the elements present in lower left part are metals while the elements in the upper right part are non metals. elements of group 13 have metallic character except the first element boron which is a metalloid, having properties intermediate between the metal and nonmetals.

General Trends In Properties Of P Block Elements These elements are found in group 13 (xiii) of the p block in the periodic table of elements. aluminum, gallium, indium, and thallium are metallic. they each have three electrons in their outermost shell (a full s orbital and one electron in the p orbital) with the valence electron configuration ns 2 np 1. The document discusses the physical and chemical properties of group 13 elements, including trends in atomic and ionic radii, ionization energy, electronegativity, density, melting points, and metallic character. Learn group 13 elements, their properties, periodic trends, and memory tips for exams. essential for jee, neet, and board preparation. In p block, the elements present in lower left part are metals while the elements in the upper right part are non metals. elements of group 13 have metallic character except the first element boron which is a metalloid, having properties intermediate between the metal and nonmetals.
Comments are closed.