Titration Lab Hcl Naoh
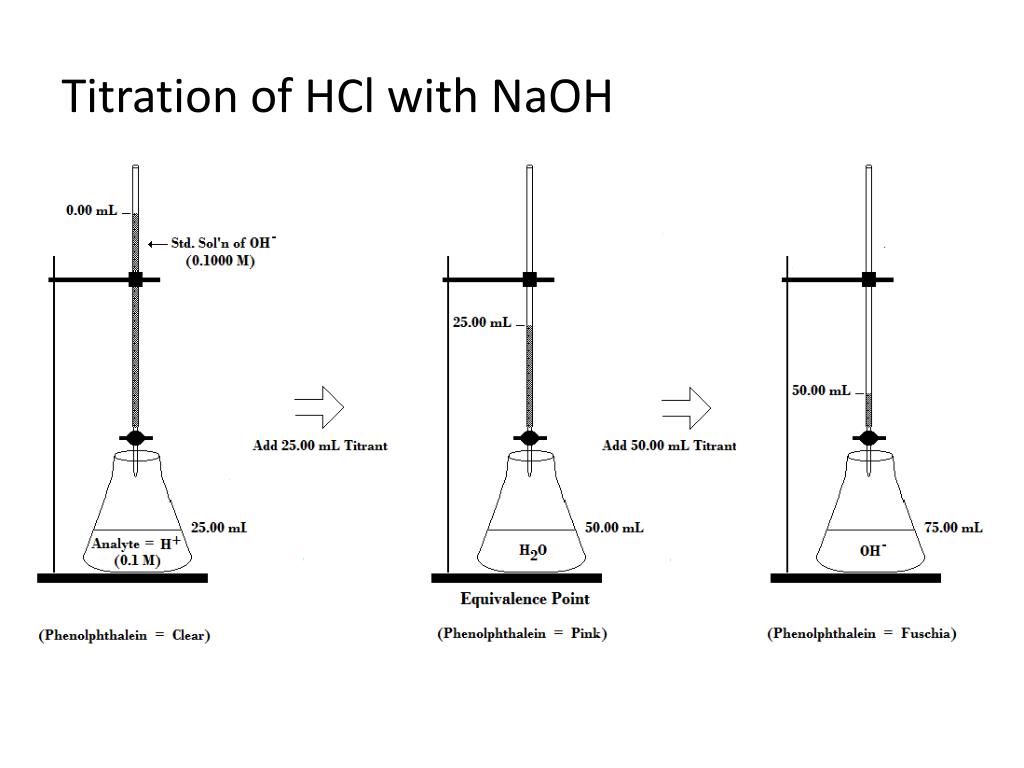
Titration Of Hcl With Naoh Simple acid base titration: determine hcl concentration using naoh. beginner friendly method for ib, igcse and gcse chemistry with step by step instructions. Lab manual for titrating hcl with naoh. includes procedure, materials, and analysis questions to determine hcl concentration.
Titration Lab With Hcl And Naoh At Jamie Heyne Blog Use this class practical to explore titration, producing the salt sodium chloride with sodium hydroxide and hydrochloric acid. includes kit list and safety instructions. To perform titration we will need titrant 0.2 m or 0.1 m sodium hydroxide solution, indicator phenolphthalein solution and some amount of distilled water to dilute hydrochloric acid sample. The document details an experiment to determine the concentration of hydrochloric acid (hcl) by titrating it with a known concentration of sodium hydroxide (naoh) using phenolphthalein as an indicator. This manual provides a brief summary to help you better understand exactly what acids and bases are as well as a detailed procedure on how to properly and safely carry out an acid base titration. in this lab we will use the base sodium hydroxide to titrate the hydrochloric acid.

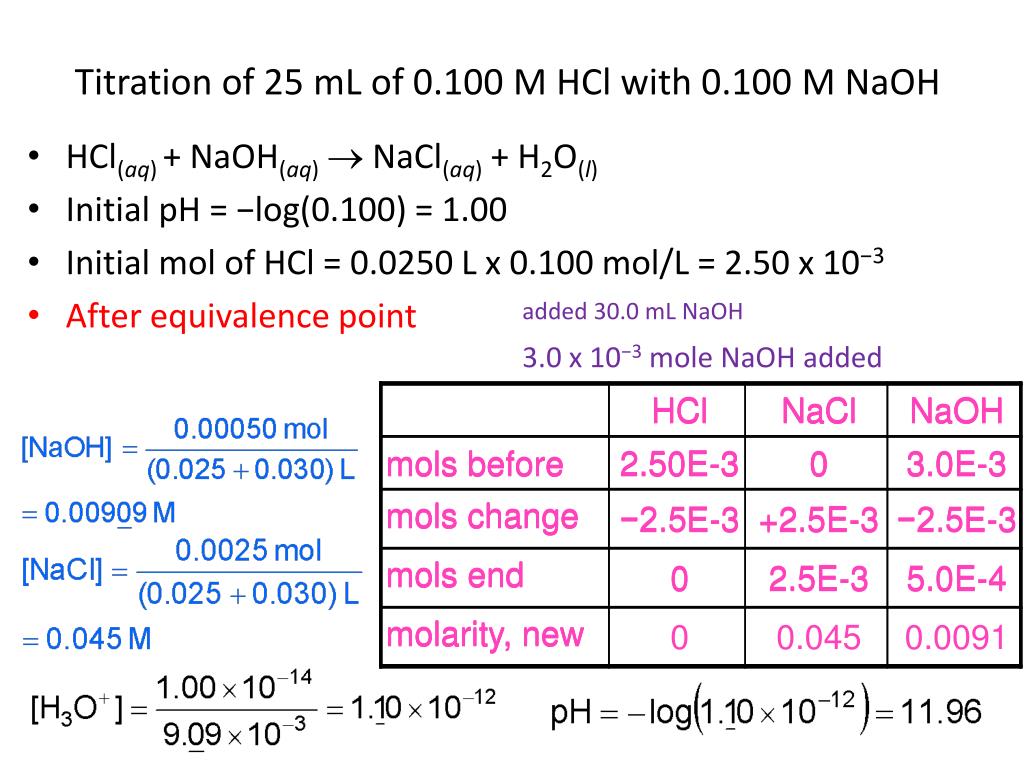
Acid Base Titration Naoh With Hcl By Fourier Education Issuu The document details an experiment to determine the concentration of hydrochloric acid (hcl) by titrating it with a known concentration of sodium hydroxide (naoh) using phenolphthalein as an indicator. This manual provides a brief summary to help you better understand exactly what acids and bases are as well as a detailed procedure on how to properly and safely carry out an acid base titration. in this lab we will use the base sodium hydroxide to titrate the hydrochloric acid. Conclusion he hydrochloric acid solution was found by stoichiometric methods. by titrating measured volumes with a strong base of kn wn concentration, the unknown molarity of the acid was determined. the process of the experiment involved preparing a calibrated bu. This lab report is about the experiment of the titration of hydrochloric acid (hcl) with sodium hydroxide (naoh). first, we draw hcl to the erlenmeyer flask and add 2 3 drop of phenolphthalein. High school lab report on titration of hydrochloric acid with sodium hydroxide. includes procedure, safety, and data analysis. Simple acid base titration: determine hcl concentration using naoh. step by step method, safety precautions and calculations for ib and igcse chemistry.
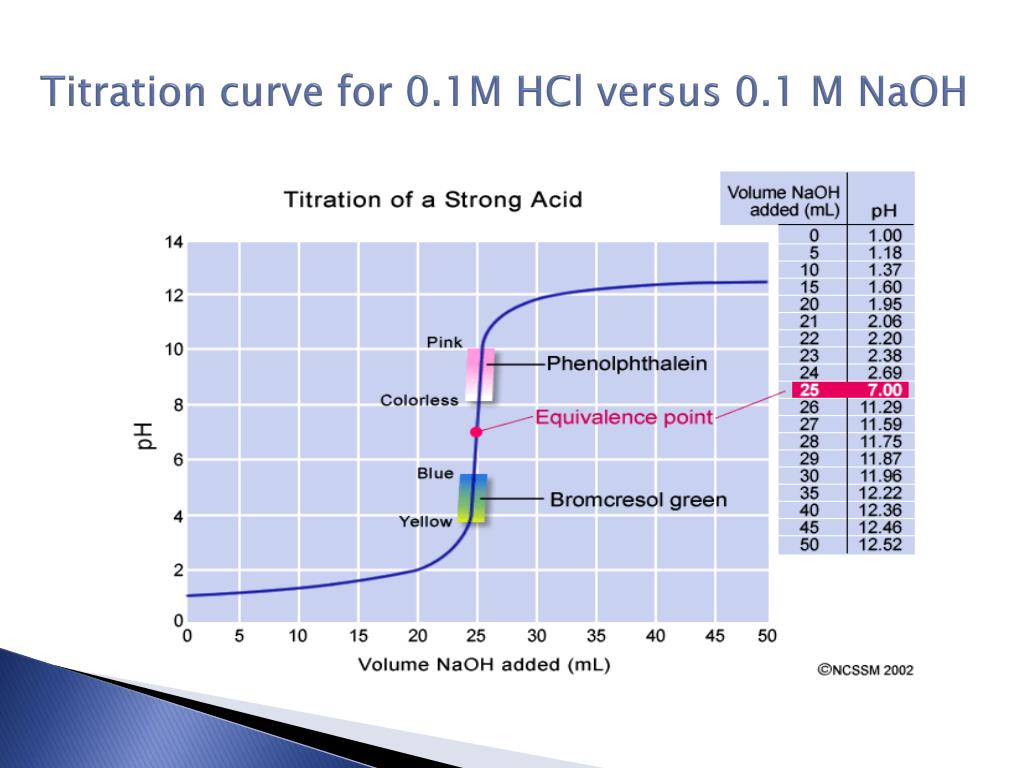
Titration Of Hcl With Naoh Conclusion he hydrochloric acid solution was found by stoichiometric methods. by titrating measured volumes with a strong base of kn wn concentration, the unknown molarity of the acid was determined. the process of the experiment involved preparing a calibrated bu. This lab report is about the experiment of the titration of hydrochloric acid (hcl) with sodium hydroxide (naoh). first, we draw hcl to the erlenmeyer flask and add 2 3 drop of phenolphthalein. High school lab report on titration of hydrochloric acid with sodium hydroxide. includes procedure, safety, and data analysis. Simple acid base titration: determine hcl concentration using naoh. step by step method, safety precautions and calculations for ib and igcse chemistry.
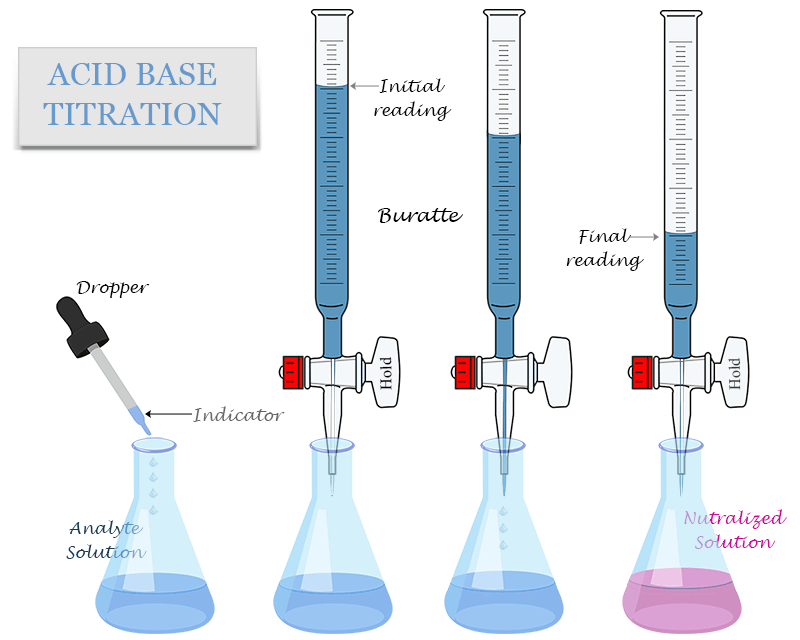
Titration Of Hcl With Naoh Ppt Titration Powerpoint Presentation High school lab report on titration of hydrochloric acid with sodium hydroxide. includes procedure, safety, and data analysis. Simple acid base titration: determine hcl concentration using naoh. step by step method, safety precautions and calculations for ib and igcse chemistry.

Acid Base Titration Lab Answers Hcl Naoh At Daniel Tilley Blog
Comments are closed.