Thermodynamic System

Thermodynamic System Qs Study A thermodynamic system is a body of matter and or radiation that can be studied using the laws of thermodynamics. learn about the types, properties, and examples of thermodynamic systems, as well as their interaction with the environment and their state of equilibrium. Thermodynamics is the study of the relations between heat, work, temperature, and energy. the laws of thermodynamics describe how the energy in a system changes and whether the system can perform useful work on its surroundings.

Thermodynamic System Energy Equilibrium Laws Learn how to define and classify thermodynamic systems, their boundaries, surroundings, and equilibrium. explore the zeroth law of thermodynamics and the concept of temperature. Thermodynamics is based on four fundamental laws — the zeroth, first, second, and third laws. each law describes a key principle of heat and energy. if two systems are each in thermal equilibrium with a third system, they are in equilibrium with each other. Learn the fundamental concepts and laws of thermodynamics, such as system, state, equilibrium, work, heat, and entropy. this pdf file covers the zeroth, first, and second laws, as well as practical applications and examples. Learn what a thermodynamic system is and how it differs from its surroundings and boundary. explore the examples and definitions of open, closed and isolated systems and their properties.
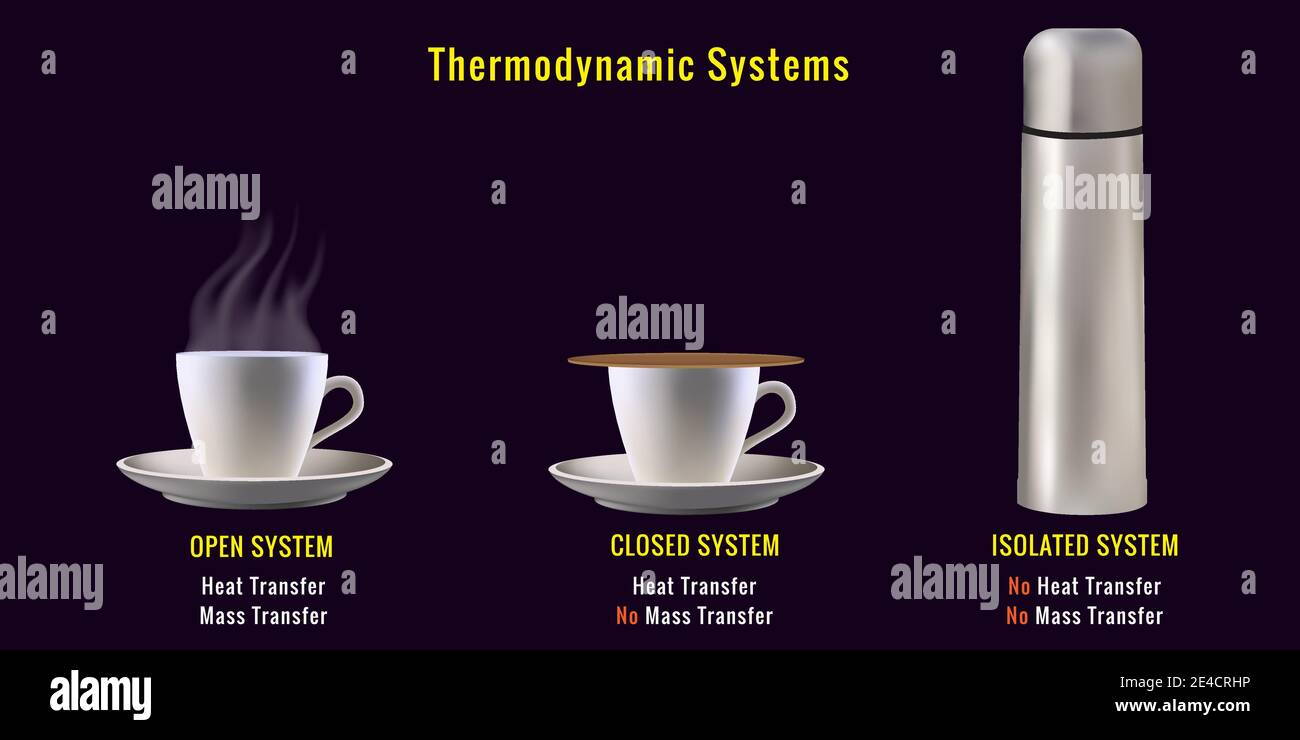
Thermodynamic System Learn the fundamental concepts and laws of thermodynamics, such as system, state, equilibrium, work, heat, and entropy. this pdf file covers the zeroth, first, and second laws, as well as practical applications and examples. Learn what a thermodynamic system is and how it differs from its surroundings and boundary. explore the examples and definitions of open, closed and isolated systems and their properties. A thermodynamic system can be described by specifying its pressure, volume, temperature, internal energy, enthalpy, and the number of moles. these parameters or variables are called thermodynamic variables. Thermodynamic systems can be defined as systems that function by utilizing the effects of heat transfer. a prominent example of a thermo system is refrigeration. Learn the definition, types, and properties of thermodynamic systems, such as temperature, pressure, and volume. explore the equation of state and the zeroth law of thermodynamics for closed systems. Learn what a thermodynamic system is and how it is classified into closed, open, and isolated systems. explore the properties of thermodynamic systems, such as intensive, extensive, state, and path functions, and their applications in engineering and science.
Comments are closed.