Thermal Assignment 1 Basic Thermodynamics Studocu
Thermodynamics Assignment 1 Pdf Course basic thermodynamics (me 209) 19documents students shared 19 documents in this course. This document outlines an assignment for the course 'thermal engineering 1' covering various topics in thermodynamics including definitions of systems, properties, processes, and laws.

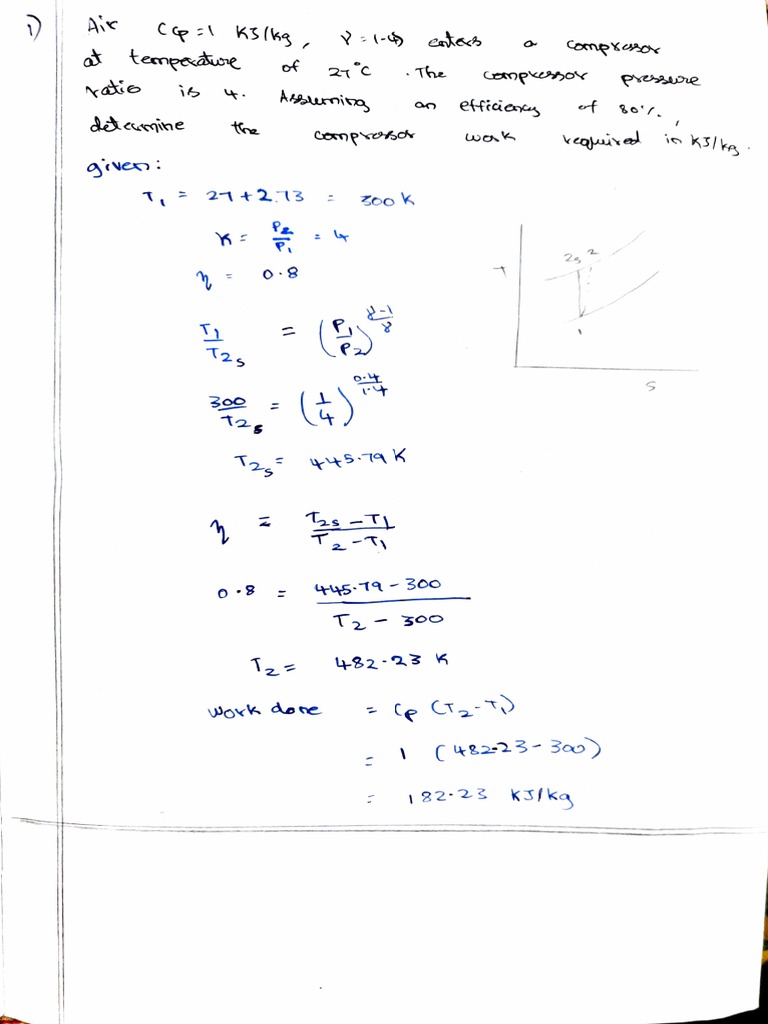
Thermal Assignment 2 Pdf Scientific Phenomena Continuum Mechanics Explore the fundamentals of thermodynamics, including approaches, processes, and properties in this comprehensive assignment on thermal science. A certain perfect gas is compressed reversibly from 1 bar, 17°c to a pressure of 5 bar in a perfectly thermally insulated cylinder, the final temperature being 77°c. Study smarter with thermodynamics notes and practice materials shared by students to help you learn, review, and stay ahead in your chemistry studies. It may seem silly that such an obvious fact is called one of the basic laws of thermodynamics. however, it cannot be concluded from the other laws of thermodynamics, and it serves as a basis for the validity of temperature measurement.

Assignment 1 Lec Thermodynamics Engg 326 0 Fall 20 23 1 Study smarter with thermodynamics notes and practice materials shared by students to help you learn, review, and stay ahead in your chemistry studies. It may seem silly that such an obvious fact is called one of the basic laws of thermodynamics. however, it cannot be concluded from the other laws of thermodynamics, and it serves as a basis for the validity of temperature measurement. It is now noted that state 1 in figure 2 3 is called a compressed liquid state because the saturation pressure for the temperature t 1 is less than p 1. at pressures below 5 mpa for water, the data are approximately equal to the saturated liquid data at the given temperature. On studocu you find all the lecture notes, summaries and study guides you need to pass your exams with better grades. This assignment explores the performance of thermodynamic power cycles, specifically comparing a recuperated reheat cycle to the conventional brayton cycle. it analyzes the impact of compressor and turbine efficiencies on thermal efficiency and specific work, highlighting design implications for gas turbine systems. This lecture notes discusses the zeroth law of thermodynamics, temperature measurement, and various types of thermometers. it explains thermal equilibrium, empirical temperature, and the relationship between pressure, volume, and temperature in thermodynamic systems, providing insights into the principles of thermal physics.

Assignment 1 Draft Version Pt1 1 Advanced Thermodynamics 1st Semester It is now noted that state 1 in figure 2 3 is called a compressed liquid state because the saturation pressure for the temperature t 1 is less than p 1. at pressures below 5 mpa for water, the data are approximately equal to the saturated liquid data at the given temperature. On studocu you find all the lecture notes, summaries and study guides you need to pass your exams with better grades. This assignment explores the performance of thermodynamic power cycles, specifically comparing a recuperated reheat cycle to the conventional brayton cycle. it analyzes the impact of compressor and turbine efficiencies on thermal efficiency and specific work, highlighting design implications for gas turbine systems. This lecture notes discusses the zeroth law of thermodynamics, temperature measurement, and various types of thermometers. it explains thermal equilibrium, empirical temperature, and the relationship between pressure, volume, and temperature in thermodynamic systems, providing insights into the principles of thermal physics.
Comments are closed.