The Post Gleevec Era
The Post Gleevec Era The success of imatinib (gleevec) is an inspiration to drug hunters. here's a post reflecting on the value it's created over the last two decades. Gleevec®, a drug used to treat chronic myelogenous leukemia (cml, a rare type of blood cancer), was among the first successful molecular medicines. this was achieved, in part, thanks to powerful scientific techniques and government efforts encouraging drug development for rare diseases.

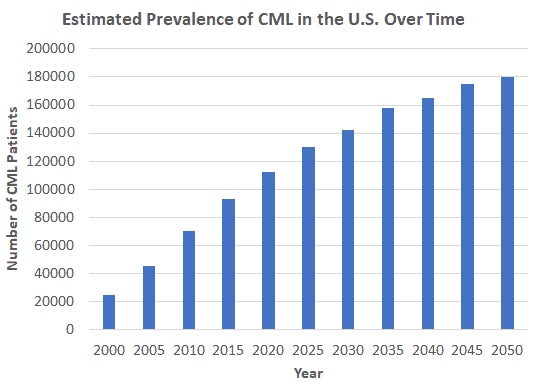
The Post Gleevec Era 🔬 the 2001 fda approval of imatinib (gleevec) consider how the 2001 approval of imatinib reshaped today’s drug regulations, a response to a breakthrough that saved chronic‑myeloid leukemia. In february 2016, a generic version of imatinib (gleevec), the groundbreaking tyrosine kinase inhibitor, was made available in the united states. imatinib has ushered in a new era of targeted oncology drug development, but has also been credited with creating a new era of drug pricing. Semantic scholar extracted view of "changes and challenges the world post gleevec (glivec)." by j. hickman et al. However, the development of gleevec (imatinib) during the early 2000s revolutionized cml treatment and marked the beginning of a new era in targeted therapy. this paper will explore gleevec’s development, mechanism of action, and its initial application to cml as well as similarly working diseases.
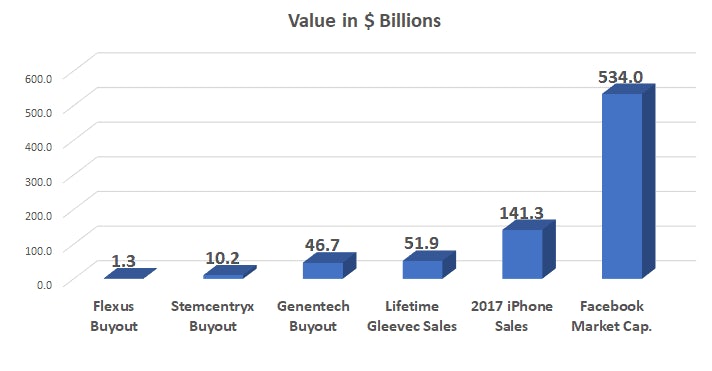
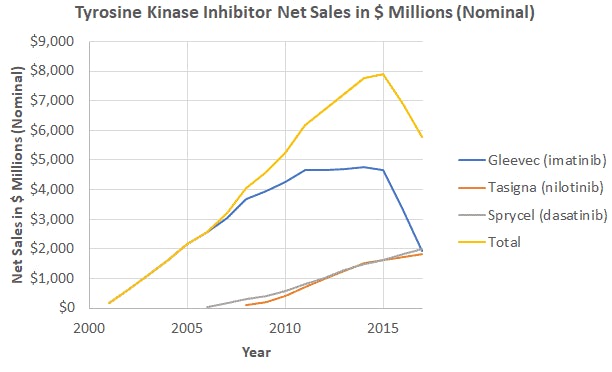
The Post Gleevec Era Semantic scholar extracted view of "changes and challenges the world post gleevec (glivec)." by j. hickman et al. However, the development of gleevec (imatinib) during the early 2000s revolutionized cml treatment and marked the beginning of a new era in targeted therapy. this paper will explore gleevec’s development, mechanism of action, and its initial application to cml as well as similarly working diseases. Extending the duration of adjuvant imatinib (gleevec) post resection from the standard 3 years to 6 years conferred a significant reduction in the risk of recurrence or death without increasing. At the conclusion of the treatment period, patients will be followed for 2 years for survival, status of response and antineoplastic treatments and quality of life. After the fda approval of gleevec, pharmaceutical labs identified “second line” treatments for cml that acted similarly to gleevec. this group includes dasatinib (sprycel) and others (there are now 5 drugs either approved or in development). During the past two decades, our understanding of the molecular pathogenesis of cancer has increased remarkably. this has fostered an explosion in the number of experimental agents and clinical.
The Post Gleevec Era Extending the duration of adjuvant imatinib (gleevec) post resection from the standard 3 years to 6 years conferred a significant reduction in the risk of recurrence or death without increasing. At the conclusion of the treatment period, patients will be followed for 2 years for survival, status of response and antineoplastic treatments and quality of life. After the fda approval of gleevec, pharmaceutical labs identified “second line” treatments for cml that acted similarly to gleevec. this group includes dasatinib (sprycel) and others (there are now 5 drugs either approved or in development). During the past two decades, our understanding of the molecular pathogenesis of cancer has increased remarkably. this has fostered an explosion in the number of experimental agents and clinical.

The Post Gleevec Era After the fda approval of gleevec, pharmaceutical labs identified “second line” treatments for cml that acted similarly to gleevec. this group includes dasatinib (sprycel) and others (there are now 5 drugs either approved or in development). During the past two decades, our understanding of the molecular pathogenesis of cancer has increased remarkably. this has fostered an explosion in the number of experimental agents and clinical.

Gleevec Structure Gleevec New Drug Approvals
Comments are closed.