The Catalyst Formula

The Catalyst Formula As can be seen from the arrhenius equation, the magnitude of the activation energy, e a, determines the value of the rate constant, k, at a given temperature and thus the overall reaction rate. catalysts provide a means of reducing e a and increasing the reaction rate. After the reaction occurs, a catalyst returns to its original state and so catalysts can be used over and over again. because it is neither a reactant nor a product, a catalyst is shown in a chemical equation by being written above the yield arrow.

The Catalyst Formula Creating Change With Our Superpowers Catalyst In autocatalysis, the catalyst is a product of the overall reaction, in contrast to all other types of catalysis considered in this article. the simplest example of autocatalysis is a reaction of type a b → 2 b, in one or in several steps. Catalytic reaction or catalysis is the process that alters the rate of a chemical reaction under the influence of a catalyst. a catalyst is a chemical substance added to a reaction to either accelerate or decelerate the reaction rate without itself undergoing any change. Catalysts do not change the overall reaction equation: the chemical formula of the reactants and the desired products is not affected by the use of a catalyst. catalysts change the reaction mechanism, which leads to a change in the rate constant | (k)| as well as the rate law. Part of the rate of formation lost by operating at lower temperatures can be recovered by using a catalyst. the net effect of the catalyst on the reaction is to cause equilibrium to be reached more rapidly.

Labflow Catalysts do not change the overall reaction equation: the chemical formula of the reactants and the desired products is not affected by the use of a catalyst. catalysts change the reaction mechanism, which leads to a change in the rate constant | (k)| as well as the rate law. Part of the rate of formation lost by operating at lower temperatures can be recovered by using a catalyst. the net effect of the catalyst on the reaction is to cause equilibrium to be reached more rapidly. A catalytic converter is a ceramic honeycomb coated with a metal catalyst mixture of platinum and rhodium or palladium. catalytic poisoning reduces the efficiency and increases the cost of catalytic converters. Because it is neither a reactant nor a product, a catalyst is shown in a chemical equation by being written above the yield arrow. a catalyst works by changing the specific way in which the reaction occurs, called its mechanism. Identify which diagram suggests the presence of a catalyst, and determine the activation energy for the catalyzed reaction: a catalyst does not affect the energy of reactant or product, so those aspects of the diagrams can be ignored; they are, as we would expect, identical in that respect. Identify which diagram suggests the presence of a catalyst, and determine the activation energy for the catalyzed reaction: a catalyst does not affect the energy of reactant or product, so those aspects of the diagrams can be ignored; they are, as we would expect, identical in that respect.
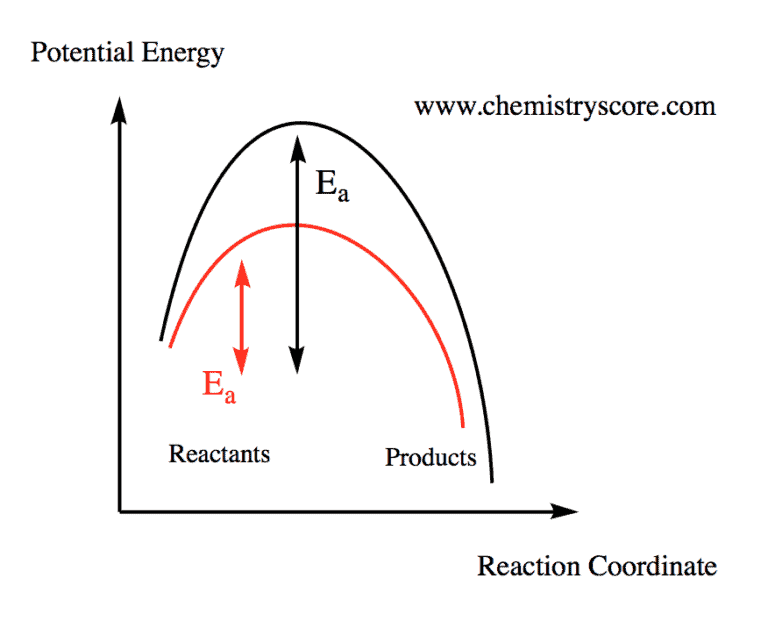
Catalyst Learn Chemistry Online Chemistryscore A catalytic converter is a ceramic honeycomb coated with a metal catalyst mixture of platinum and rhodium or palladium. catalytic poisoning reduces the efficiency and increases the cost of catalytic converters. Because it is neither a reactant nor a product, a catalyst is shown in a chemical equation by being written above the yield arrow. a catalyst works by changing the specific way in which the reaction occurs, called its mechanism. Identify which diagram suggests the presence of a catalyst, and determine the activation energy for the catalyzed reaction: a catalyst does not affect the energy of reactant or product, so those aspects of the diagrams can be ignored; they are, as we would expect, identical in that respect. Identify which diagram suggests the presence of a catalyst, and determine the activation energy for the catalyzed reaction: a catalyst does not affect the energy of reactant or product, so those aspects of the diagrams can be ignored; they are, as we would expect, identical in that respect.
Comments are closed.