The 2025 Fda Dietary Supplement Compliance Program Update Rosemarie

Fda Regulations On Dietary Supplements Pdf Dietary Supplements The fda has overhauled its dietary supplement compliance program, and those who don't adapt quickly may find themselves facing costly inspections, product holds, or worse. The fda’s actions in 2025 reflect a renewed commitment to the safety and efficacy of dietary supplements. with structural reorganization and a stronger focus on new ingredients, the agency seeks to ensure that consumers have access to safe, accurately labeled products.

Fda Dietary Supplement Inspections And Compliance Support In ninety minutes, learn exactly what inspectors will look for in 2025: new quality control requirements critical labeling assessment criteria manufacturer vs. distributor responsibilities. Fda issues guidance documents containing nonbinding recommendations to help industry understand and comply with all regulations. The fda is enhancing its inspection processes in the dietary supplement industry, focusing on compliance and self regulation to improve food safety. new initiatives include the use of ai for data analysis and unannounced inspections, along with a push for transparency in ingredient safety. Food labeling: notification procedures for statements on dietary supplements—21 cfr 101.93.

The 2025 Fda Dietary Supplement Compliance Program Update Rosemarie The fda is enhancing its inspection processes in the dietary supplement industry, focusing on compliance and self regulation to improve food safety. new initiatives include the use of ai for data analysis and unannounced inspections, along with a push for transparency in ingredient safety. Food labeling: notification procedures for statements on dietary supplements—21 cfr 101.93. This article provides a detailed overview of fda’s dietary supplement compliance system, highlighting key regulatory requirements, manufacturing obligations, and inspection strategies for 2026. Each quarter, i provide updates on notable fda warning letters and enforcement trends. understanding enforcement trends is essential for being a savvy marketer, regulatory affairs professional or dietary supplement executive. Understand the 2025 fda rules for supplements, differences between usp monographs and gmp compliance, and their impact. Below are short summaries of significant recent developments regarding fda’s regulation of food and dietary supplements, including labeling issues and product safety considerations.

Fda Dietary Supplement Program Updates This article provides a detailed overview of fda’s dietary supplement compliance system, highlighting key regulatory requirements, manufacturing obligations, and inspection strategies for 2026. Each quarter, i provide updates on notable fda warning letters and enforcement trends. understanding enforcement trends is essential for being a savvy marketer, regulatory affairs professional or dietary supplement executive. Understand the 2025 fda rules for supplements, differences between usp monographs and gmp compliance, and their impact. Below are short summaries of significant recent developments regarding fda’s regulation of food and dietary supplements, including labeling issues and product safety considerations.
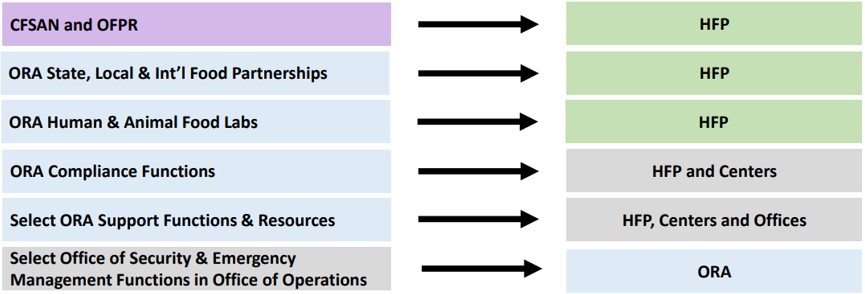
Fda Dietary Supplement Program Updates Understand the 2025 fda rules for supplements, differences between usp monographs and gmp compliance, and their impact. Below are short summaries of significant recent developments regarding fda’s regulation of food and dietary supplements, including labeling issues and product safety considerations.
Comments are closed.