Temperature And Heat Flow Activity

Temperatures Mass Flow Heat Flow And Heat Flow Over Return When they are heated, they move faster and when they are cooled, they move slower. for example, if you put a room temperature metal spoon into a hot liquid like soup or hot chocolate, the metal gets hotter. Maintaining deep body temperature requires constant metabolic activity as a heat source (endothermy), as well as control of metabolic heat flow from deep tissues to the environment.
80 944 Heat Flow Images Stock Photos Vectors Shutterstock Discuss with the students about the density difference arising from temperature difference between water in the beaker and the conical flask. after some time, float an ice cube on water in the beaker and observe what happens. Along with elite athletes, recreational competitors also face performance and potential health challenges due to extreme heat exposure. these challenges are associated with the rise in core temperature and loss of body water related to exercising in the heat. Students' eyes are opened to the value of creative, expressive and succinct visual presentation of data, findings and concepts. student pairs design, redesign and perform simple experiments to test the differences in thermal conductivity (heat flow) through different media (foil and thin steel). In this activity, students can analyze a heating curve to understand how heat flows through a substance. a heating curve is a graph that shows how temperature changes over time as a substance is heated.

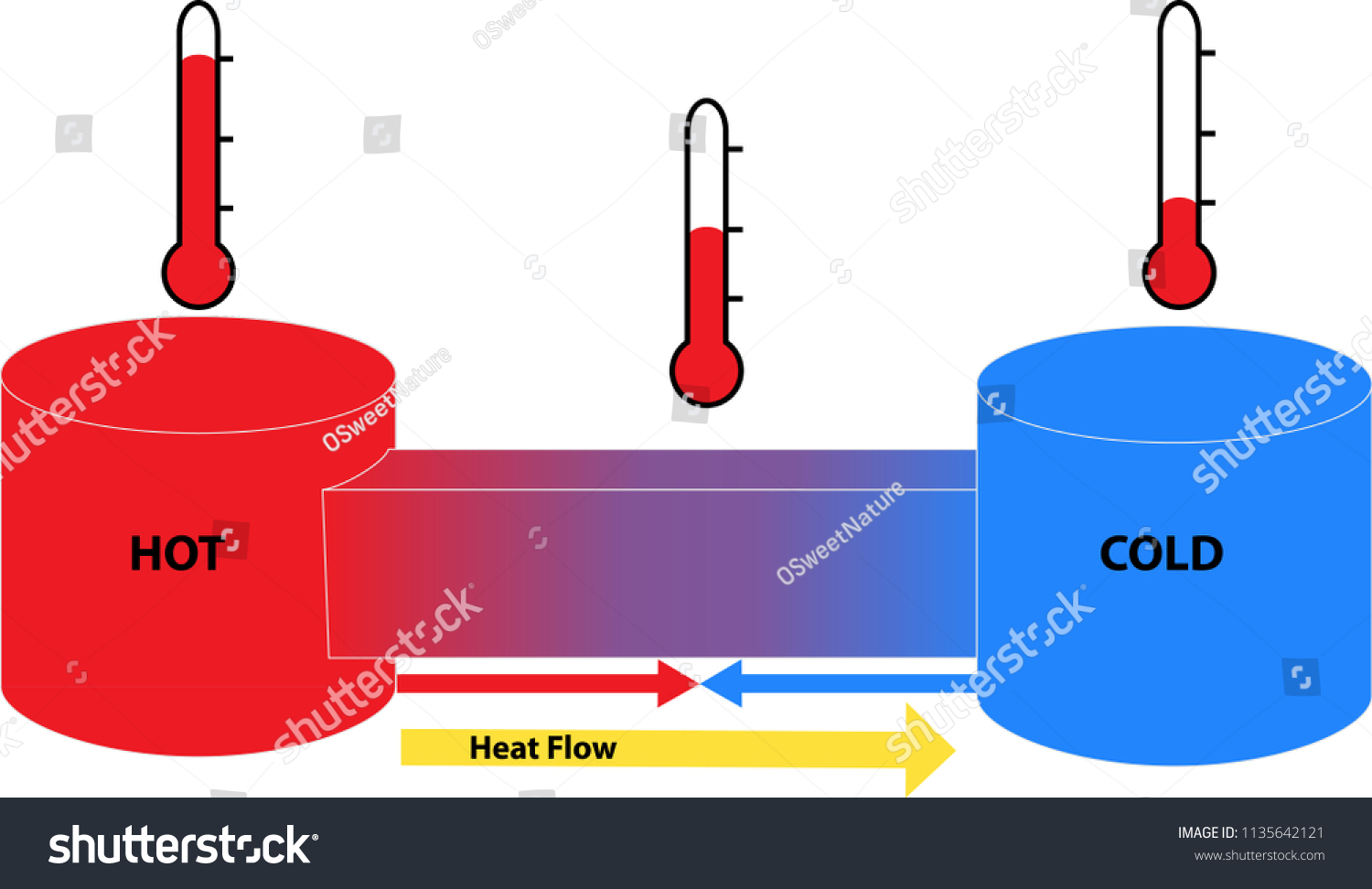
Heat Flow As A Function Of Temperature Download Scientific Diagram Students' eyes are opened to the value of creative, expressive and succinct visual presentation of data, findings and concepts. student pairs design, redesign and perform simple experiments to test the differences in thermal conductivity (heat flow) through different media (foil and thin steel). In this activity, students can analyze a heating curve to understand how heat flows through a substance. a heating curve is a graph that shows how temperature changes over time as a substance is heated. Students will understand the concept of heat as the movement of thermal energy from warmer to cooler objects. students will watch a short video clip about a snowman in the summer. students will hypothesize about the flow of heat. Heat is the flow of energy from one object to another. this flow of energy is caused by a difference in temperature. the transfer of heat can change temperature, as can work, another kind of energy transfer that is central to thermodynamics. This article explores the physiological processes of thermoregulation, the anatomical structures involved, and their significance in adapting to environmental and activity induced temperature changes. Use the equation for heat transfer q = mcΔt to express the heat lost by the aluminum pan in terms of the mass of the pan, the specific heat of aluminum, the initial temperature of the pan, and the final temperature: qhot = ma1ca1 ⎛⎝tf − 150 °c⎞⎠.
Comments are closed.