Table 2 From Cd19 Cd22 Dual Targeted Car T Therapy Active In Relapsed
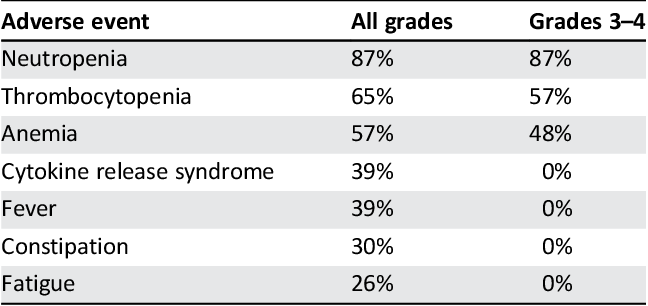
Table 1 From Cd19 Cd22 Dual Targeted Car T Therapy Active In Relapsed In summary, preliminary findings from alexander highlight the potential safety and efficacy benefits of dual‐targeted anti‐cd19 cd22 car t‐cell therapy in patients with relapsed refractory dlbcl. Chimeric antigen receptor (car) t cell therapies that target either cd19 or cd22 alone have potent antilymphoma effects. however, antigen escape–mediated relapse often occurs. car t cells targeting both cd19 and cd22 may overcome this limitation.

Table 2 From Cd19 Cd22 Dual Targeted Car T Therapy Active In Relapsed Our meta analysis demonstrated that the cd22 cd19 dual targeting car t cell strategy has high efficiency with tolerable adverse effects in b cell malignancies. To address antigen escape and expression heterogeneity, we developed four cd19 cd22 car constructs with different configurations, designated as tancar t1, tancar t2, loopcar t1, and loopcar t2. Bone marrow transplantation sequential cd19 cd22 car t cell therapy following asct shows improved efficacy versus car t alone in relapsed refractory large b cell lymphoma. In this study, we developed a fully human, bivalent loop bi car t targeting both cd19 and cd22 (ct120). we then conducted an open label, single center, single arm phase i ii trial to assess its efficacy and safety in patients with relapsed or refractory non hodgkin lymphoma (r r nhl).

Clinical Trials Of Dual Targeted Car T Cell Therapy In Mm Download Bone marrow transplantation sequential cd19 cd22 car t cell therapy following asct shows improved efficacy versus car t alone in relapsed refractory large b cell lymphoma. In this study, we developed a fully human, bivalent loop bi car t targeting both cd19 and cd22 (ct120). we then conducted an open label, single center, single arm phase i ii trial to assess its efficacy and safety in patients with relapsed or refractory non hodgkin lymphoma (r r nhl). Dual targeting car t cells cotransduced with cd19 and cd22 cars were used to treat 12 patients with relapsed refractory all with a 1 year efs of 60%. at a median follow up of 8.7 months, there were no cases of antigen negative relapse, suggesting this approach may prevent antigen escape. In this study, we engineered cd19 cd22 bs loop car t cells with an enhanced targeting efficacy for cd22 and assessed their safety and effectiveness in patients with relapsed refractory diffuse large b cell lymphoma. Relapses frequently occur following cd19 directed chimeric antigen receptor (car) t cell treatment for relapsed or refractory b cell acute lymphocytic leukaemia in children. we aimed to assess the activity and safety of sequential cd19 directed and cd22 directed car t cell treatments. Because antigen loss and lack of car t cell persistence are the leading causes of progressive disease following single antigen targeting, we evaluated cd22 cd19 dual targeting.
Comments are closed.