Supercritical Carbon Dioxide Assignment Point

Supercritical Carbon Dioxide Pdf Supercritical Fluid Carbon Dioxide While conventional power plant cycles produce power from turbines using water or steam as the working fluid, supercritical carbon dioxide (sco2) cycles use co2 that is in a supercritical state—at a temperature and pressure above its critical point where liquid and gas phases are not distinguishable. Supercritical carbon dioxide (sco 2[disputed – discuss]) is a fluid state of carbon dioxide where it is held at or above its critical temperature and critical pressure.
Supercritical Carbon Dioxide Assignment Point This behavior can be attributed to the unique characteristics of scco 2 at conditions above its critical point. in the vicinity of the critical point, elevated temperature and pressure conditions enhance the thermal motion of co2 molecules while diminishing intermolecular forces. Planetary environments with supercritical fluids, particularly supercritical carbon dioxide, exist, even on earth (below the ocean floor), on venus, and likely on super earth type exoplanets. these planetary environments may present a possible habitat for exotic life. All above mentioned characteristics make supercritical co2 highly attractive to be used as a "green solvent" and have led to multiple practical applications in different industries. Supercritical co 2 occurs when carbon dioxide is heated and compressed beyond its critical point. at this threshold, the boundaries between liquid and gas phases disappear.

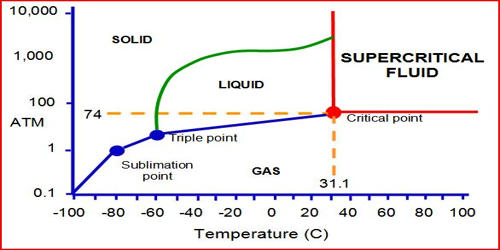
Super Critical Carbon Dioxide Modelling Pdf Supercritical Fluid All above mentioned characteristics make supercritical co2 highly attractive to be used as a "green solvent" and have led to multiple practical applications in different industries. Supercritical co 2 occurs when carbon dioxide is heated and compressed beyond its critical point. at this threshold, the boundaries between liquid and gas phases disappear. Supercritical carbon dioxide has been used in the decaffeination of coffee beans since the 1980s, it is preferred over other methods of decaffeination as it leaves the flavour intact, removes the highest percentage (97% 99%) of caffeine, and the residue is lost easily to the environment. Supercritical fluids (scfs) are compounds at a temperature and pressure beyond their critical point. carbon dioxide is the most widely used scf owing to its reasonable critical point (304.1 k and 7.38 mpa), low cost, inert and non toxic character. As shown by this pressure temperature phase diagram, carbon dioxide is a supercritical fluid above its critical point of 301.1 k and 73.8 bar. a supercritical fluid is any substance at a temperature and pressure above its thermodynamic critical point. The gradual phase transition of carbon dioxide from a single phase supercritical state through the critical point to a two phase subcritical state. (the view cell shown here has a glass tube inserted in the cell).
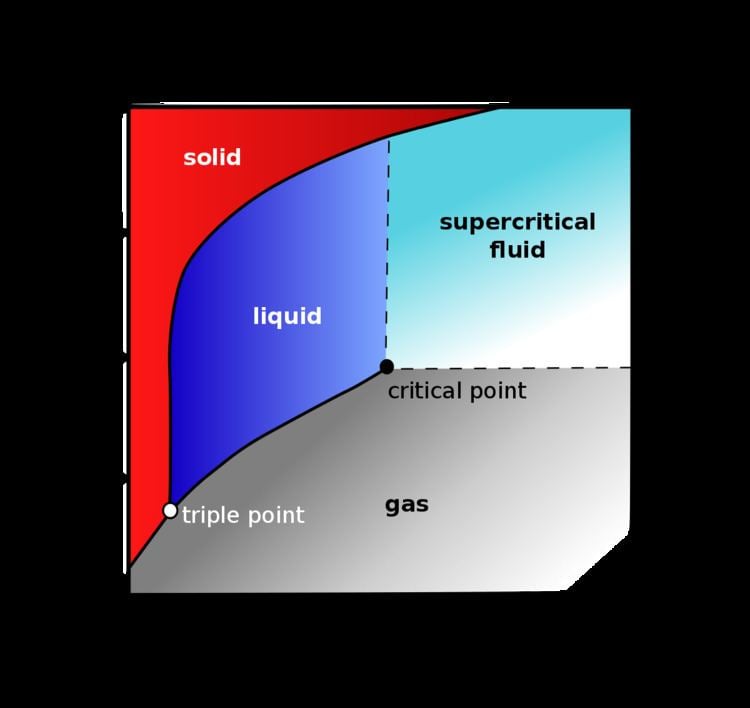
Supercritical Carbon Dioxide Alchetron The Free Social Encyclopedia Supercritical carbon dioxide has been used in the decaffeination of coffee beans since the 1980s, it is preferred over other methods of decaffeination as it leaves the flavour intact, removes the highest percentage (97% 99%) of caffeine, and the residue is lost easily to the environment. Supercritical fluids (scfs) are compounds at a temperature and pressure beyond their critical point. carbon dioxide is the most widely used scf owing to its reasonable critical point (304.1 k and 7.38 mpa), low cost, inert and non toxic character. As shown by this pressure temperature phase diagram, carbon dioxide is a supercritical fluid above its critical point of 301.1 k and 73.8 bar. a supercritical fluid is any substance at a temperature and pressure above its thermodynamic critical point. The gradual phase transition of carbon dioxide from a single phase supercritical state through the critical point to a two phase subcritical state. (the view cell shown here has a glass tube inserted in the cell).
Comments are closed.