Sunrise 4 New Breakthroughs In Bladder Cancer Treatment And Biomarkers Andrea Necchi At Esmo2025

Biomarkers For Bladder Cancer In this esmo 2025 highlight, dr. andrea necchi — medical oncologist, associate professor at vita salute san raffaele university, and director of genitourinar. At the esmo 2025 annual meeting, dr. necchi and colleagues reported the efficacy and safety from the primary analysis of sunrise 4, and urinary (utdna) and circulating tumor dna (ctdna) minimal residual disease as biomarkers of outcomes agnostic to treatment.
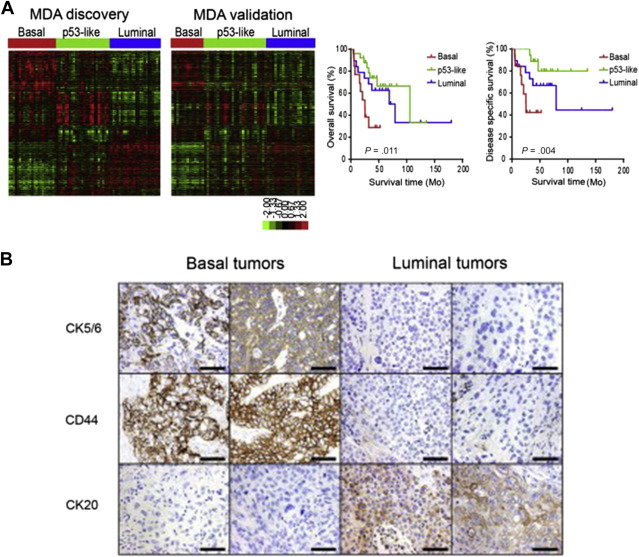
Emerging Bladder Cancer Biomarkers And Targets Of Therapy Abdominal Key There were no new safety signals for tar 200, cetrelimab, or the combination. serious treatment related adverse events were reported in 13.9% of patients in the combination arm and 5.2% of patients in the monotherapy arm. Exploratory analyses highlight urinary and circulating tumor dna as promising biomarkers for residual disease and treatment outcomes in this setting. this interview took place at the european society for medical oncology (esmo) 2025 congress in berlin, germany. During the esmo congress, necchi shared findings from the primary analysis of the study, as well as data from exploratory biomarker analyses of utdna and ctdna mrd. In an interview, andrea necchi, md, università vita salute san raffaele, discusses the data from sunrise 4 presented at the 2025 esmo congress. the findings are particularly relevant for patients scheduled for radical cystectomy who are ineligible for or refuse standard cisplatin based chemotherapy.
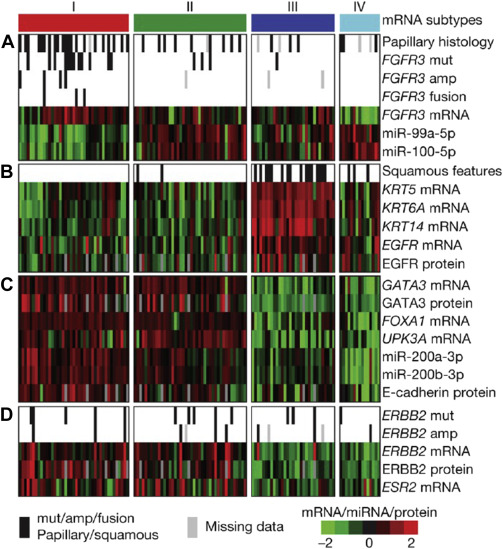
Emerging Bladder Cancer Biomarkers And Targets Of Therapy Abdominal Key During the esmo congress, necchi shared findings from the primary analysis of the study, as well as data from exploratory biomarker analyses of utdna and ctdna mrd. In an interview, andrea necchi, md, università vita salute san raffaele, discusses the data from sunrise 4 presented at the 2025 esmo congress. the findings are particularly relevant for patients scheduled for radical cystectomy who are ineligible for or refuse standard cisplatin based chemotherapy. Johnson & johnson announced results from the primary analysis of its ongoing phase 2b sunrise 4 study, which evaluated the combination of inlexzo (gemcitabine intravesical system) and intravenous cetrelimab in patients with muscle invasive bladder. 1 necchi, a. et al., neoadjuvant gemcitabine intravesical system (tar 200) cetrelimab or cetrelimab alone in patients with muscle invasive bladder cancer: sunrise 4 primary analysis and biomarker results. Andrea necchi, md, discusses results from the phase 2 sr 4 trial evaluating neoadjuvant tar 200 plus cetrelimab compared to cetrelimab alone in patients with muscle invasive bladder cancer ineligible for or declined platinum based chemotherapy. Here, we report efficacy, safety, and exploratory biomarker outcomes from the primary analysis of sunrise 4. the study protocol was designed by the sponsor, johnson & johnson.

Bladder Cancer Biomarkers Johnson & johnson announced results from the primary analysis of its ongoing phase 2b sunrise 4 study, which evaluated the combination of inlexzo (gemcitabine intravesical system) and intravenous cetrelimab in patients with muscle invasive bladder. 1 necchi, a. et al., neoadjuvant gemcitabine intravesical system (tar 200) cetrelimab or cetrelimab alone in patients with muscle invasive bladder cancer: sunrise 4 primary analysis and biomarker results. Andrea necchi, md, discusses results from the phase 2 sr 4 trial evaluating neoadjuvant tar 200 plus cetrelimab compared to cetrelimab alone in patients with muscle invasive bladder cancer ineligible for or declined platinum based chemotherapy. Here, we report efficacy, safety, and exploratory biomarker outcomes from the primary analysis of sunrise 4. the study protocol was designed by the sponsor, johnson & johnson.

Pdf Identifying Novel Biomarkers Associated With Bladder Cancer Andrea necchi, md, discusses results from the phase 2 sr 4 trial evaluating neoadjuvant tar 200 plus cetrelimab compared to cetrelimab alone in patients with muscle invasive bladder cancer ineligible for or declined platinum based chemotherapy. Here, we report efficacy, safety, and exploratory biomarker outcomes from the primary analysis of sunrise 4. the study protocol was designed by the sponsor, johnson & johnson.
Breakthroughs In Bladder Cancer Treatment Revolutionize Survival Rates
Comments are closed.