Substitution Reaction Alkane
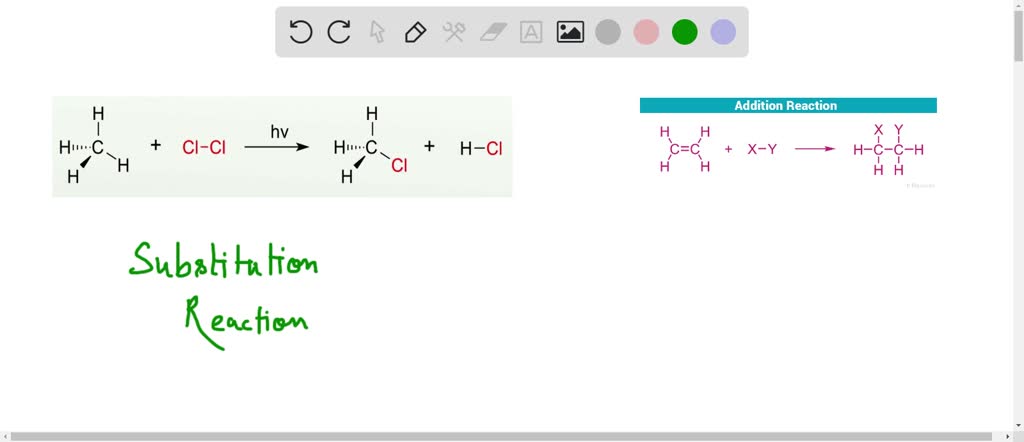
Solved Why Does A Substitution Reaction Involving An Alkane And A Define substitution reaction in alkanes with detailed explanations, mechanisms, applications, and key concepts for cambridge igcse chemistry. Unlike the complex transformations of combustion, the halogenation of an alkane appears to be a simple substitution reaction in which a c h bond is broken and a new c x bond is formed.

Halogenation Reaction Of Alkane A Free Radical Substitution Reaction In a substitution reaction, a reacting species is substituted (swapped) for a bonded species in a compound. alkanes can react with halogens in the presence of ultraviolet (uv) light. a halogen is substituted for a hydrogen atom in the alkane to form a halogenoalkane. the process happens in stages. Learn about chemical reactivity and reactions of alkanes including substitution reactions, catalytic cracking, and combustion of alkanes. In this article, we will discuss all things related to substitution reaction i.e., its definition, and examples including alkane substitution, nucleophilic, electrophilic, free radical as well as organomatelic substitution. Substitution reactions represent one of the most important transformations of alkanes. in these reactions, hydrogen atoms are replaced by halogen atoms (such as chlorine or bromine) through a process called halogenation.
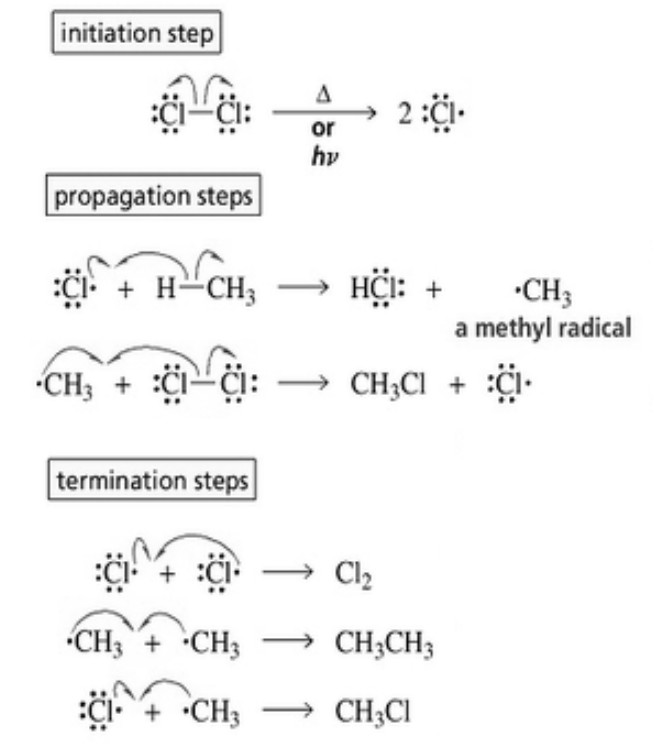
Substitution Reaction Alkane In this article, we will discuss all things related to substitution reaction i.e., its definition, and examples including alkane substitution, nucleophilic, electrophilic, free radical as well as organomatelic substitution. Substitution reactions represent one of the most important transformations of alkanes. in these reactions, hydrogen atoms are replaced by halogen atoms (such as chlorine or bromine) through a process called halogenation. Alkanes contain only c−c and c−h σ bonds. since σ bonds are quite strong bonds, alkanes are generally inert towards acids, bases, oxidising and reducing agents. however, they undergo the following reactions under certain conditions : in substitution reactions, a hydrogen of a hydrocarbon is replaced by an atom or a group of atoms. In a substitution reaction, one functional group in a compound is replaced by another, allowing chemists to modify the molecule’s properties and reactivity. these reactions are significant for synthesising primary haloalkanes and primary alcohols. In this article, we will learn about substitution reactions of alkanes. we will discuss the substitution reactions of alkanes through examples. Amines 2 2 3 4 . ethers. thiols (mercaptans) sulfides.
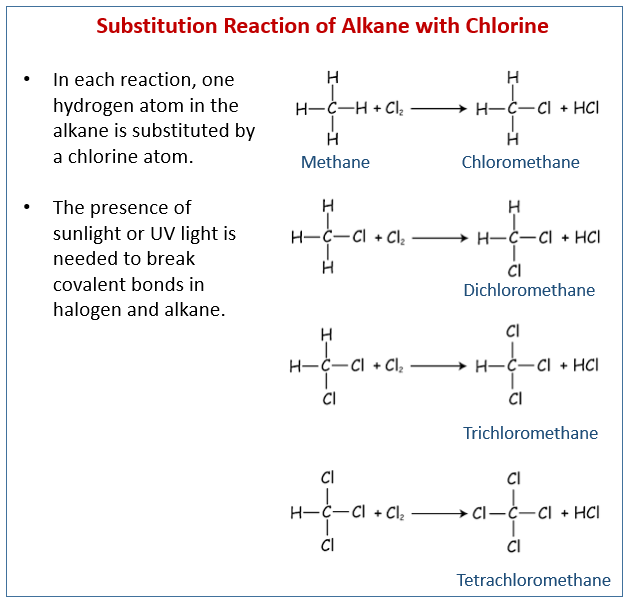
Substitution Reaction Alkane Alkanes contain only c−c and c−h σ bonds. since σ bonds are quite strong bonds, alkanes are generally inert towards acids, bases, oxidising and reducing agents. however, they undergo the following reactions under certain conditions : in substitution reactions, a hydrogen of a hydrocarbon is replaced by an atom or a group of atoms. In a substitution reaction, one functional group in a compound is replaced by another, allowing chemists to modify the molecule’s properties and reactivity. these reactions are significant for synthesising primary haloalkanes and primary alcohols. In this article, we will learn about substitution reactions of alkanes. we will discuss the substitution reactions of alkanes through examples. Amines 2 2 3 4 . ethers. thiols (mercaptans) sulfides.

Substitution Reaction Alkane In this article, we will learn about substitution reactions of alkanes. we will discuss the substitution reactions of alkanes through examples. Amines 2 2 3 4 . ethers. thiols (mercaptans) sulfides.
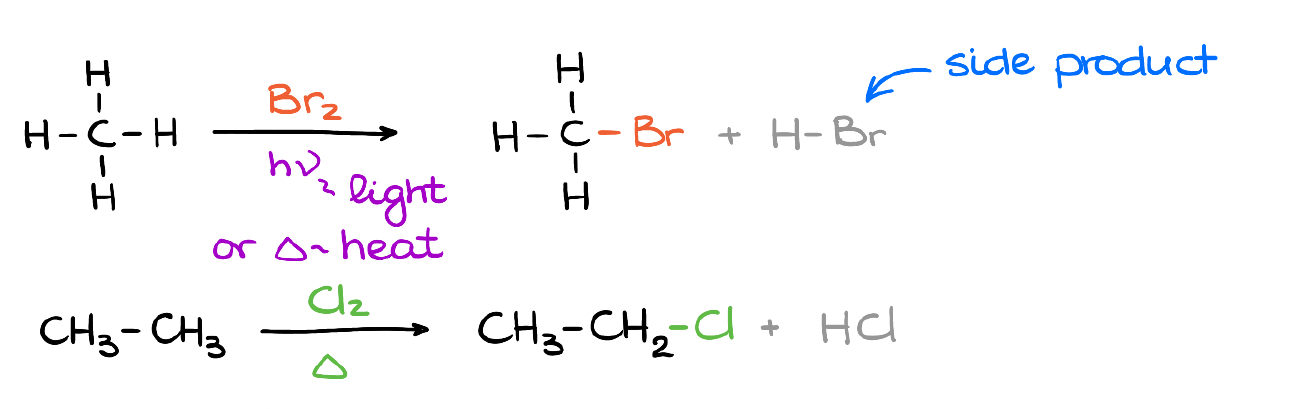
Substitution Reaction Alkane
Comments are closed.